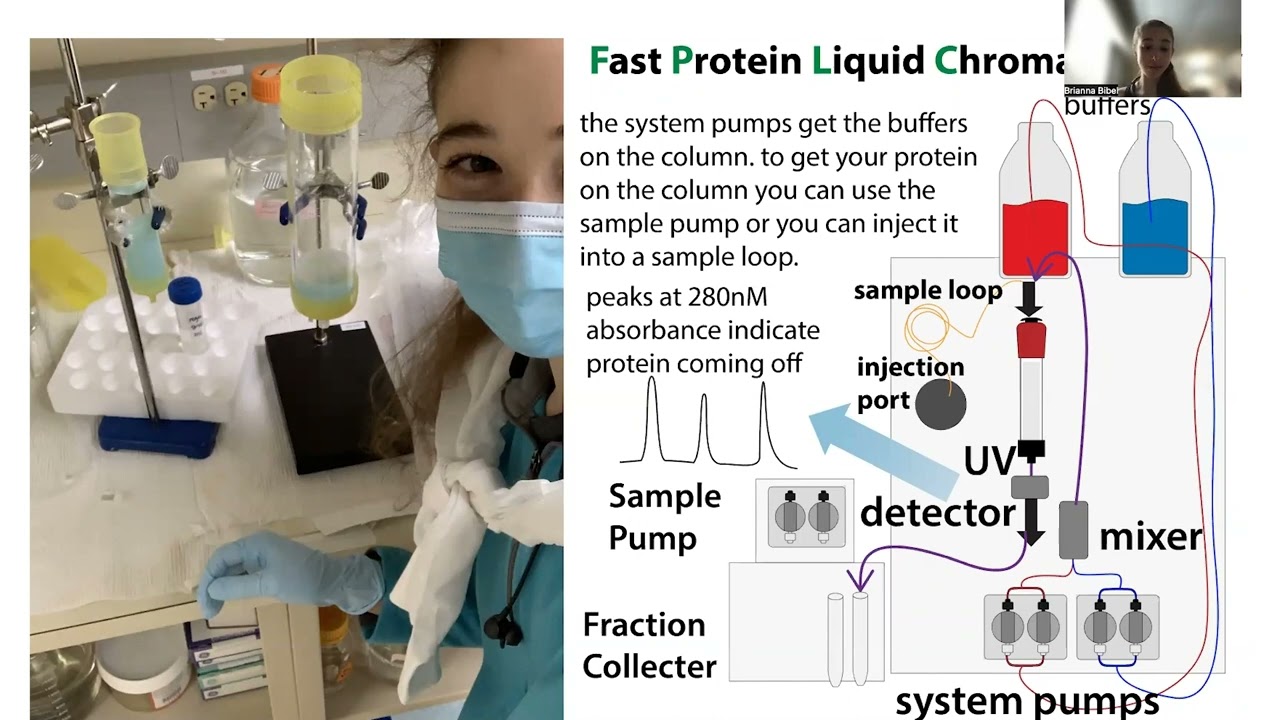
Techniques for buffer exchange (Desalting columns, Dialysis & Diafiltration): Pros, cons & going-ons
Автор: the bumbling biochemist
Загружено: 2026-02-20
Просмотров: 465
Описание:
The 3 D’s of buffer exchange! There are 3 main techniques for buffer exchange of proteins (or nucleic acids): Desalting (via gel filtration/size exclusion chromatography), Dialysis, and Diafiltration (typically using a spin concentrator)
https://thebumblingbiochemist.com/365...
Here’s the gist:
Desalting (Size Exclusion Chromatography/Gel Filtration (typically using “desalting columns”): The resin has tiny pores – proteins, DNA, RNA, etc. can’t get into any pores, so they flow right through, coming out with the buffer that was in the pores already. Meanwhile, the buffer they come in with is trapped in the pores.
Dialysis: Uses a membrane pouch with movement of small things reversibly through via diffusion. Put your protein solution in a membrane pouch & put it in a big bath of liquid to exchange with. Little stuff diffuses in AND out, but your protein’s stuck. Since it relies on diffusion, the inner & outer buffers will reach equilibrium, so you need to use a large bath volume that you replace midway through.
Diafiltration (typically using spin concentrators): Uses a membrane with forced one-way movement of small things through. You start by concentrating your protein, pulling the old buffer through (e.g., by centrifuging). Then you dilute it in new buffer, pull that through (concentrating again), dilute it again in new buffer (concentrating again), dilute it again . . . 2-5 times. Then you can resuspend in the final volume you want.
Pros & Cons of different buffer exchange techniques:
Desalting:
Pros:
Can be done quickly, with large volumes
Lots of formats depending on needs: FPLC columns, gravity flow columns, spin columns, all of various sizes
Great for small volumes too
Cons:
Sample doesn’t get concentrated, might get diluted
Dialysis:
Pros:
Simple
No fancy equipment needed
Can cut membrane tubing to custom sizes best for your sample
Cons:
SLOW (typically done overnight, often in a cold room)
Needs LOTS of buffer (200-400X sample volume), with exchanging partway through
Tubing can be finicky and might accidentally spill when setting it up (so I recommend setting up over a clean beaker)
Diafiltration:
Pros:
You control final volume
Faster than dialysis
Uses much less buffer than dialysis
Good for small volumes
Cons:
Your protein might “crash out” (precipitate) at high concentrations
Your protein might aggregate on the membrane
You have to do multiple (3-5) cycles of concentrating/diluting/concentrating/diluting, which can be time-consuming
More on desalting columns: https://bit.ly/desalt_buffer_exchange
More on dialysis: https://bit.ly/proteindialysis ; YouTube: • Protein dialysis in biochemistry - theory ...
More on spin concentrators: https://bit.ly/spinconcentrators & • Practical tips for using spin concentrator...
More on avoiding crashing out: blog: https://bit.ly/crashingout ; YouTube: • Practical tips for preventing proteins fro...
For more practical protein-purification posts (and background/theory). http://bit.ly/proteinpurificationtech
more about all sorts of things: #365DaysOfScience All (with topics listed) 👉 http://bit.ly/2OllAB0 or search blog: http://thebumblingbiochemist.com
#scicomm #biochemistry #molecularbiology #biology #sciencelife #science #realtimechem
Повторяем попытку...

Доступные форматы для скачивания:
Скачать видео
-
Информация по загрузке: