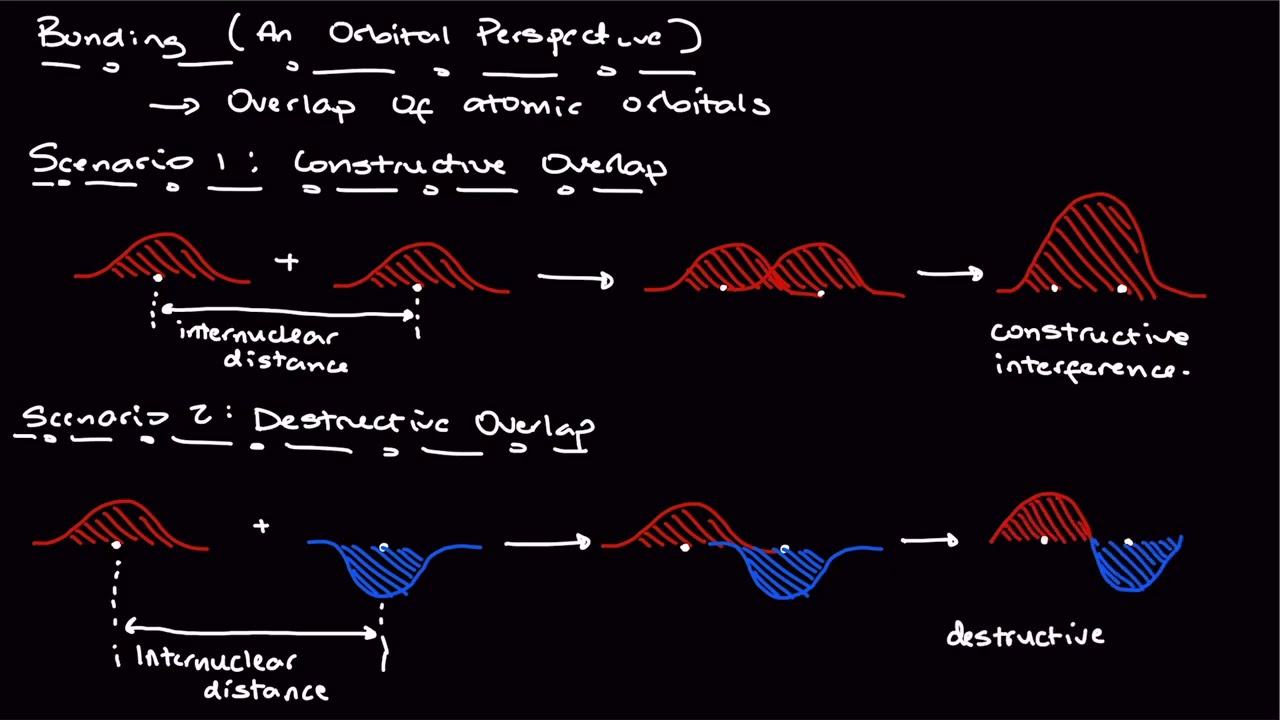
Electrocyclic Reactions
Автор: Chemistry with Prof. Ogba
Загружено: 2026-01-28
Просмотров: 56
Описание:
The video introduces electrocyclic reactions. It focuses on the cyclization of conjugated polyenes and how molecular orbital theory explains their stereochemical outcomes.
By the end of the video, viewers should be able to:
– Draw and identify the products of electrocyclic reactions, considering both the cyclization and ring-opening parts.
– Draw curved arrow mechanisms.
– Draw transition state structures for electrocyclic reactions.
– Use molecular orbital theory to explain the observed stereochemical outcomes of electrocyclic reactions.
– Understand the concept of disrotatory and conrotatory motion.
– Predict stereochemical outcomes based on thermal versus photochemical conditions.
Timestamps
00:00 - Introduction and Learning Goals
00:43 - Definition of Electrocyclic Reactions
01:01 - Example of a Cyclization Reaction (Hexatriene)
01:22 - Curved Arrow Mechanism and Transition State
02:18 - Considering the Stereochemistry of Electrocyclic Reactions
03:38 - Origin of Selectivity and Molecular Orbital Theory
04:54 - Predicting Stereochemical Outcomes: Focus on the HOMO Orbital
05:47 - Molecular Orbital Picture for a 6π System under Thermal Conditions
07:39 - Disrotatory Motion (Symmetrical HOMO)
10:25 - Conrotatory Motion (Anti-Symmetrical HOMO)
12:27 - Summary of Symmetrical vs. Anti-Symmetrical Orbitals
13:11 - Molecular Orbital Picture for an 8π System under Thermal Conditions
18:31 - Thermal vs. Photochemical Conditions and the Change in Product Stereochemistry
19:25 - How Photochemical Conditions Change the HOMO Symmetry
22:59 - Practice Problems (4π Systems)
25:20 - Conclusion and Summary of Learning Goals
Повторяем попытку...

Доступные форматы для скачивания:
Скачать видео
-
Информация по загрузке: