Molecular Orbital Theory
Автор: Chemistry with Prof. Ogba
Загружено: 2026-01-24
Просмотров: 96
Описание:
The video provides a review of molecular orbital theory.
By the end of the video, viewers should be able to:
– Draw and identify atomic orbitals.
– Define a node in atomic or molecular orbitals.
– Identify the number and location of nodes in an orbital picture.
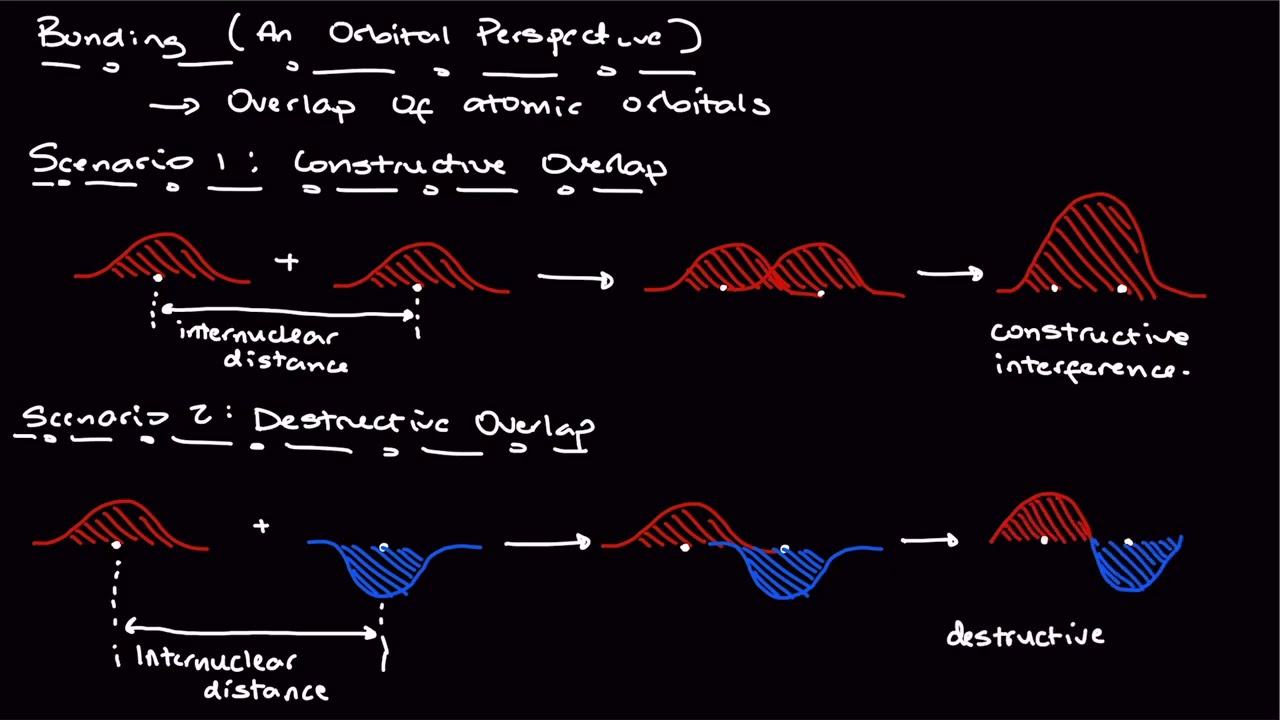
– Predict the molecular orbital picture resulting from constructive and destructive overlap of two orbitals.
– Identify the Highest Occupied Molecular Orbital (HOMO) and the Lowest Unoccupied Molecular Orbital (LUMO).
– Draw molecular orbital diagrams for sigma and pi overlaps in any molecule.
Timestamps
00:00 - Introduction and Learning Goals
01:20 - Understanding Electrons as Waves
04:38 - The Quantum Mechanical Model and Atomic Orbitals
11:39 - Molecular Orbitals and Overlap
14:18 - Molecular Orbital Diagram for H2
22:20 - Example: CH3Cl and Carbon-Chlorine σ Overlap
26:38 - Molecular Orbital Diagrams for π Systems
30:16 - Molecular Orbital Diagrams for Extended π Systems
36:55 - Conclusion and Summary of Key Concepts
#MolecularOrbitalTheory #OrganicChemistry #ChemistryWithProfOgba #HOMOLUMO
Повторяем попытку...
Доступные форматы для скачивания:
Скачать видео
-
Информация по загрузке:









![1 A.M Study Session 📚 [lofi hip hop]](https://imager.clipsaver.ru/lTRiuFIWV54/max.jpg)








