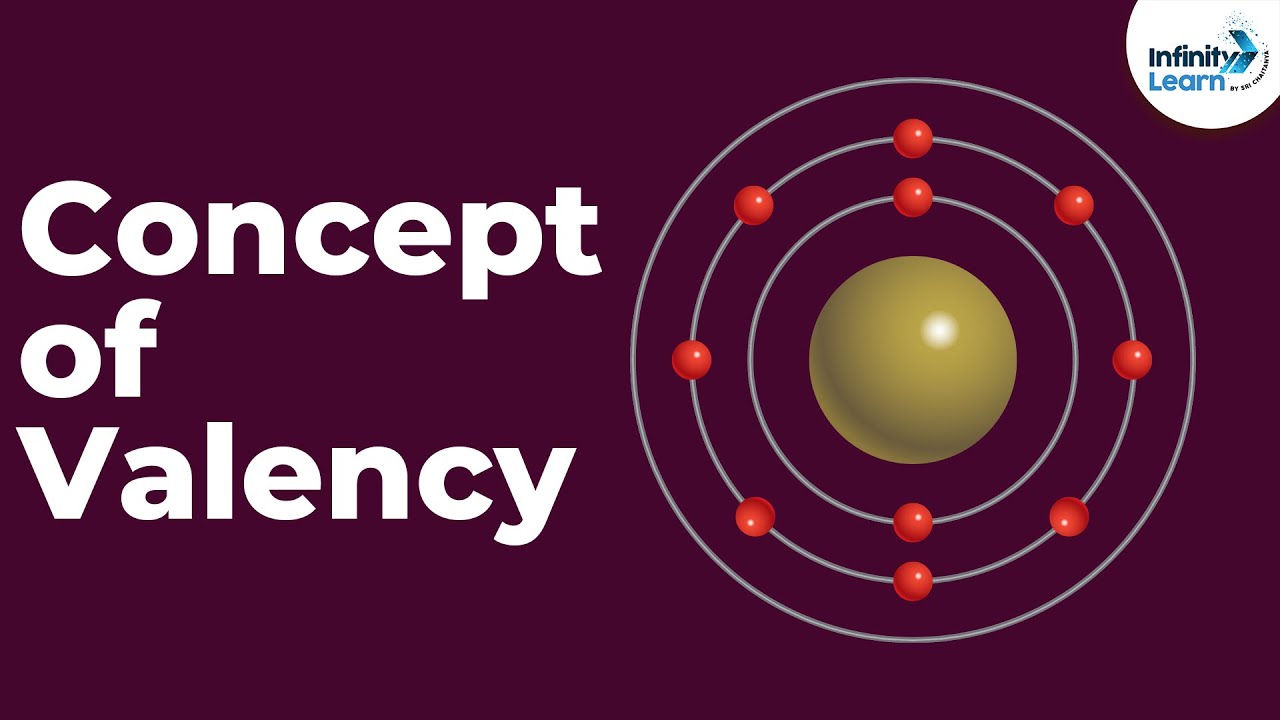
Which is larger? Ca or Ca 2+ (Calcium atom or Calcium ion)
Автор: Wayne Breslyn (Dr. B.)
Загружено: 2023-06-26
Просмотров: 7273
Описание:
The calcium ion (Ca2+) is smaller than the neutral calcium atom (Ca) primarily because of changes in the electron configuration and the resulting effective nuclear charge.
In a neutral calcium atom, there are 20 electrons distributed across different energy levels or electron shells. The outermost electron shell of the calcium atom is the fourth shell, which contains two electrons.
When calcium loses two electrons to form the Ca2+ ion, it becomes positively charged. The loss of these two electrons affects the electron configuration and the overall balance of positive and negative charges in the ion.
In the Ca2+ ion, there are only 18 electrons remaining. Since there are now more protons (20) than electrons (18) in the ion, the effective nuclear charge experienced by the remaining electrons increases. The greater positive charge attracts the electrons more strongly towards the nucleus, causing the electron cloud to be pulled in closer. This results in a decrease in the ionic radius of Ca2+ compared to the atomic radius of the neutral calcium atom.
Therefore, the reduction in the number of electrons and the increased effective nuclear charge contribute to the smaller size of the Ca2+ ion compared to the neutral Ca atom.
Based on these general trends we can say that Ca is larger than Ca 2+ .
Image from: https://en.wikipedia.org/wiki/File:20...
Повторяем попытку...

Доступные форматы для скачивания:
Скачать видео
-
Информация по загрузке:













![Эффект Джанибекова [Veritasium]](https://imager.clipsaver.ru/N9HlQ-XVnFk/max.jpg)