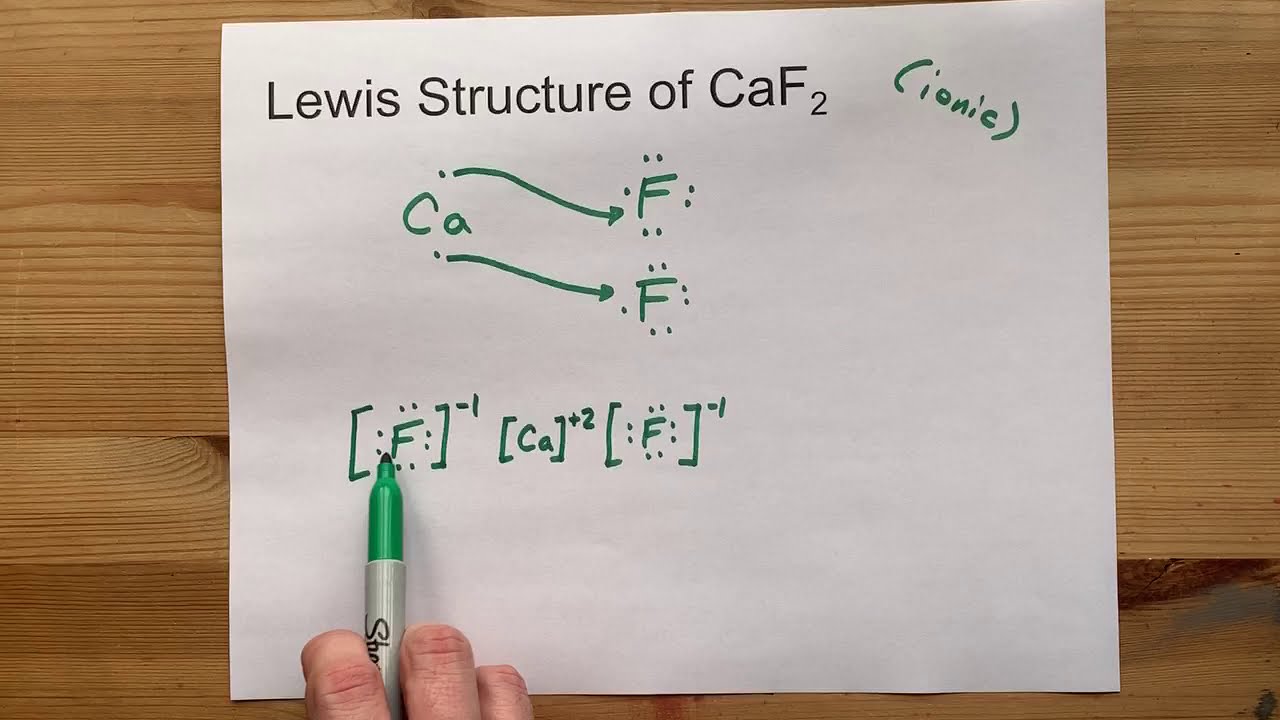
Draw the Lewis Structure of CaF2 (calcium fluoride)
Автор: chemistNATE
Загружено: 2020-08-22
Просмотров: 34101
Описание:
Calcium Fluoride (CaF2 or F2Ca) is an ionic compound.
One calcium atom loses two electrons, so it becomes +2 charge
Two fluorine atoms gain those two electrons (one electron each) and become a -1 charge each.
These ions are held together with an ionic bond, and we use square brackets to show the charge in the Lewis Structure.
Check me out: http://www.chemistnate.com
Повторяем попытку...
Доступные форматы для скачивания:
Скачать видео
-
Информация по загрузке:















![Эффект Джанибекова [Veritasium]](https://imager.clipsaver.ru/N9HlQ-XVnFk/max.jpg)



