CH322 - Alzheimer’s Disease Part 2: Proteostasis Failure, Autophagy, and Lysosome Dysfunction
Автор: Patricia Flatt
Загружено: 2026-03-08
Просмотров: 13
Описание:
In this second lecture on Alzheimer’s disease, we examine how failures in cellular protein quality control systems contribute to neurodegeneration.
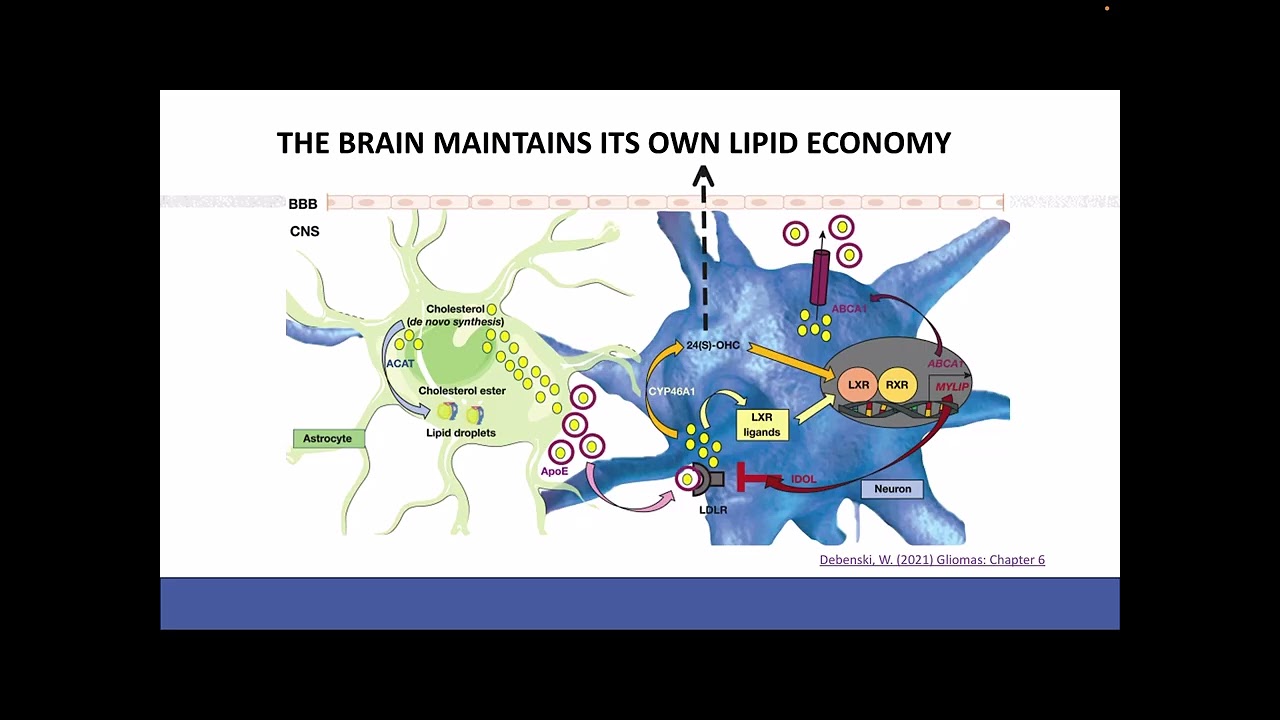
Healthy neurons rely on an integrated proteostasis network to maintain proper protein folding, trafficking, recycling, and degradation. This network includes the ubiquitin–proteasome system, endosomes, lysosomes, and autophagy pathways, which together remove damaged proteins and maintain cellular homeostasis.
In Alzheimer’s disease, these systems begin to break down. Disruptions in endosomal trafficking, lysosomal degradation, and autophagy lead to the accumulation of toxic proteins such as amyloid-β and tau, increasing cellular stress and ultimately contributing to neuronal death.
Topics covered in this lecture include:
• The proteostasis network and its role in neuronal health
• Ubiquitination and proteasome-mediated protein degradation
• Endosome trafficking and receptor recycling
• The role of lysosomes in cellular degradation
• Autophagy and autophagosome formation in neurons
• Endosome–lysosome dysfunction in Alzheimer’s disease
• Failure of lysosomal acidification and protein degradation
• Autophagy–lysosome fusion defects and accumulation of toxic proteins
• How proteostasis failure contributes to amyloid and tau pathology
Understanding these cellular mechanisms provides an important foundation for exploring emerging therapeutic strategies aimed at restoring protein clearance and cellular recycling pathways in neurodegenerative disease.
This lecture is intended for students studying biochemistry, medicinal chemistry, neuroscience, pharmacology, and neurodegenerative disease biology.
Повторяем попытку...

Доступные форматы для скачивания:
Скачать видео
-
Информация по загрузке:













![ЛИМФА – эти 3 действия настолько эффективны, что даже ПУГАЮТ! [Бесплатно]](https://imager.clipsaver.ru/fBUrNm8GhNY/max.jpg)


