RAM III: Atomic Mass Units
Автор: AtomicSchool
Загружено: 2020-05-26
Просмотров: 7094
Описание:
Description: An atom weighs about a ten trillion TRILLIONTH of a kilogram. This video describes the units that are just the right size to measure the incredibly tiny mass of atoms- the unified atomic mass unit (u), or Dalton (Da). It also explores how John Dalton originally imagined it over two hundred years ago.
You may be interested in this related video on the same topic explaining Dalton’s original experimental method to do this: • Chemistry Tutorial: Atomic & Molecular Mass
Transcript: In earlier videos, we lined up soccer players from the lightest to the heaviest, and pretended they were like different atoms, with each Player Number like an Atomic Number, and each Player Mass like an Atomic Mass. Now let’s change from a Soccer Team to an Atom Team.
In 1865, English scientist John Newlands listed the elements from lightest to heaviest, along with their place numbers, and he also claimed that this revealed a repeating pattern in the elements.
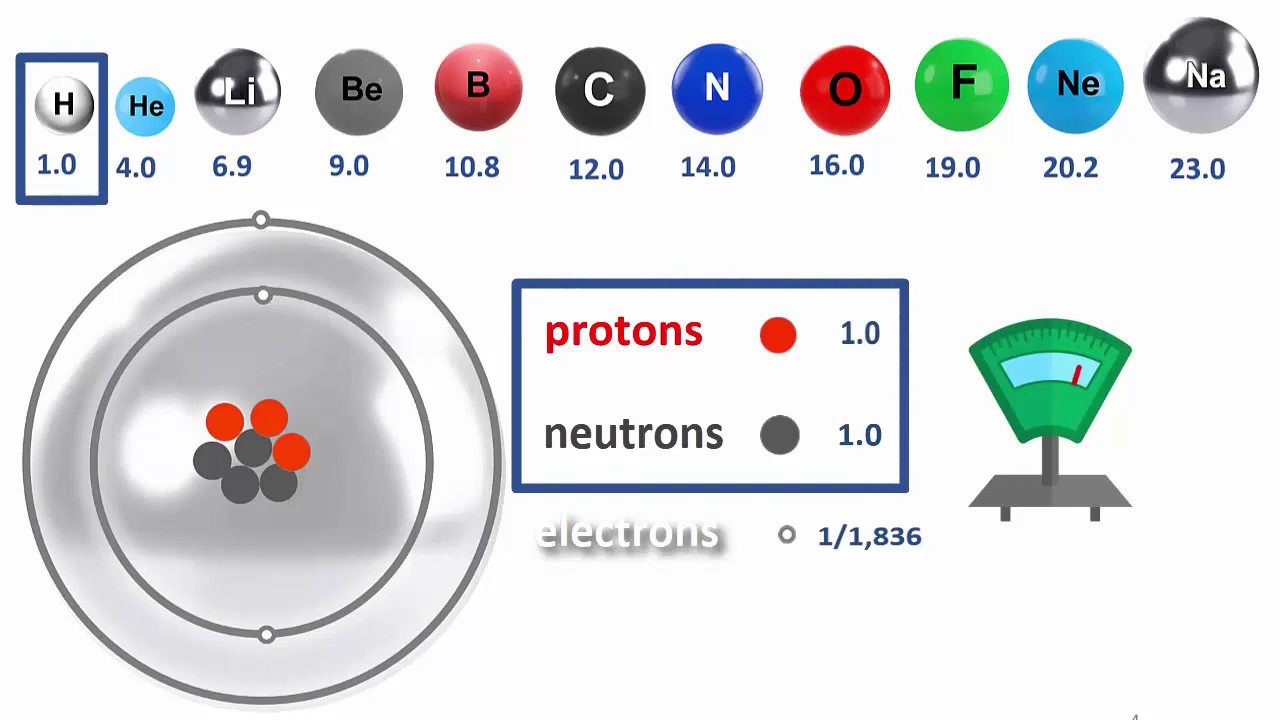
This is how other scientists reacted. But he was onto something. But listing atoms by their masses was about to reveal their secrets, so he was onto something. How could scientists make a tiny enough scale to measure the masses of atoms, which are incredibly light? A single lithium atom weighs about a ten trillion trillionth of a kilogram! That’s 24 zeros after the decimal point. Kilograms are way too big to conveniently measure atoms. Scientists needed a new tiny scale.
In 1803, another English scientist John Dalton was the first to create a mass scale especially for atoms. To keep it simple, he didn’t want any fractions less than 1, which would’ve been a pain in the … elbow. By giving an atomic mass of 1 to the lightest atom, all the other atoms were automatically made bigger than 1. Which’d be simpler. What’s the lightest kind of atom? Hydrogen. So it’s mass is made equal to 1. But 1 what?
This new scale needed a new unit, and scientists called it the “unified atomic mass unit”. shortened to amu, and then further to just u. They also gave it a second name, the Dalton, shortened to Da, which is a bit confusing. This means that the mass of one hydrogen atom is 1 u, or 1 Dalton; your choice. But I like u, so we’ll stick with that here.
We used this mass scale in kg to weigh our soccer players. But It’s way too clunky to weigh atoms, and Dalton used a different method which you can check out in a video linked in the description. But we’ll pretend that this scale can measure atoms in u. Let’s do it.
You can see that lithium is now 6.9 u. Before we said it was a ten trillion trillionth of a kg. Does that mean that its mass has changed with this new scale? No, 6.9 u is the same a ten trillion trillionth of a kg. The same mass is just being measured in different units. Just like you get different numbers if you weigh yourself in kilograms or pounds.
We can now line up the atoms in order of their masses, and put their place positions in, too. Which will take first place, and which will take last place?
Hydrogen is first, obviously, and sodium is last here. Now we have 2 rows of numbers, each atoms’ mass on the bottom, and place number on the top.
You know scientists call the atom’s place number their Atomic Number. And they call their actual mass their Atomic Mass, not surprisingly.
This reminds us of the Player Number and Player Mass of our Soccer team.
Notice that Atomic Numbers are consecutive whole numbers- like the Player Numbers - whereas the Atomic Masses have decimal places- like the Player Masses.
Also Atomic Numbers increase smoothly from one to the next, whereas the Atomic Masses increase unevenly. For example, Hydrogen to Helium’s jumps from 1.0 to 4.0- a gap of 3 u. Helium to Lithium from 4.0 to 6.9, a gap of 2.9, Lithium to Beryllium 6.9 to 9.0, a gap of 2.1, and so on.
We’ll find out why that happens later.
In the next video we’ll show how scientists tweaked the definition of Atomic Mass by comparing atoms’ masses to carbon instead of hydrogen, and also how scientists killed u in order to create Relative Atomic Mass. It’s a neat trick!
Повторяем попытку...

Доступные форматы для скачивания:
Скачать видео
-
Информация по загрузке:

![What is Atomic Mass & Mass Defect on the Periodic Table? - [1-2-9]](https://imager.clipsaver.ru/XLCupmUZimY/max.jpg)