Скачать
ammonium ions as acids
Автор: Chem Help ASAP
Загружено: 2019-06-17
Просмотров: 933
Описание:
directory of Chem Help ASAP videos: https://www.chemhelpasap.com/youtube/
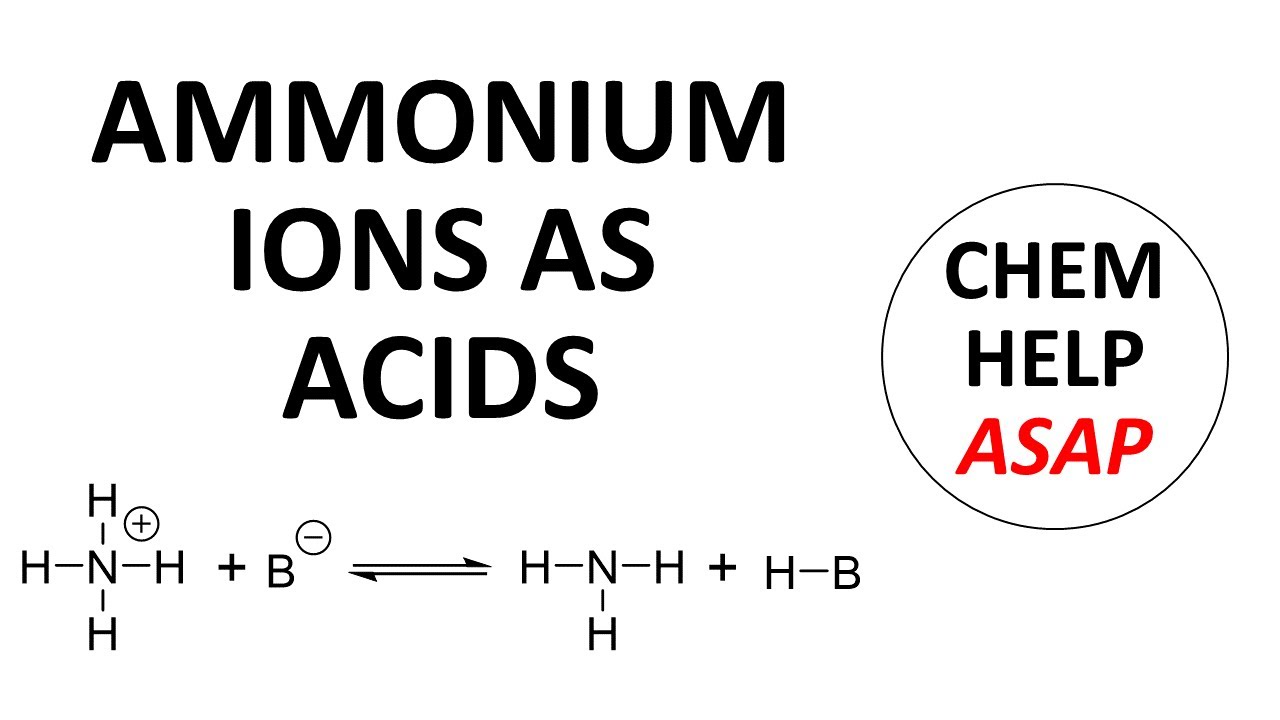
In general, acids in acid-base reactions are neutral with H-A being a typical representation of an acid. When H-A loses a proton, it forms A- as the conjugate base. In some cases, however, the acid starts with a positive charge and loses a proton to form a neutral conjugate base. One example is an ammonium ion, NH4+. Another, perhaps more common, example is hydronium ion, H3O+. While positively charged acids are less common, they are still regularly encountered.
Не удается загрузить Youtube-плеер. Проверьте блокировку Youtube в вашей сети.
Повторяем попытку...
Повторяем попытку...
Доступные форматы для скачивания:
Скачать видео
-
Информация по загрузке: