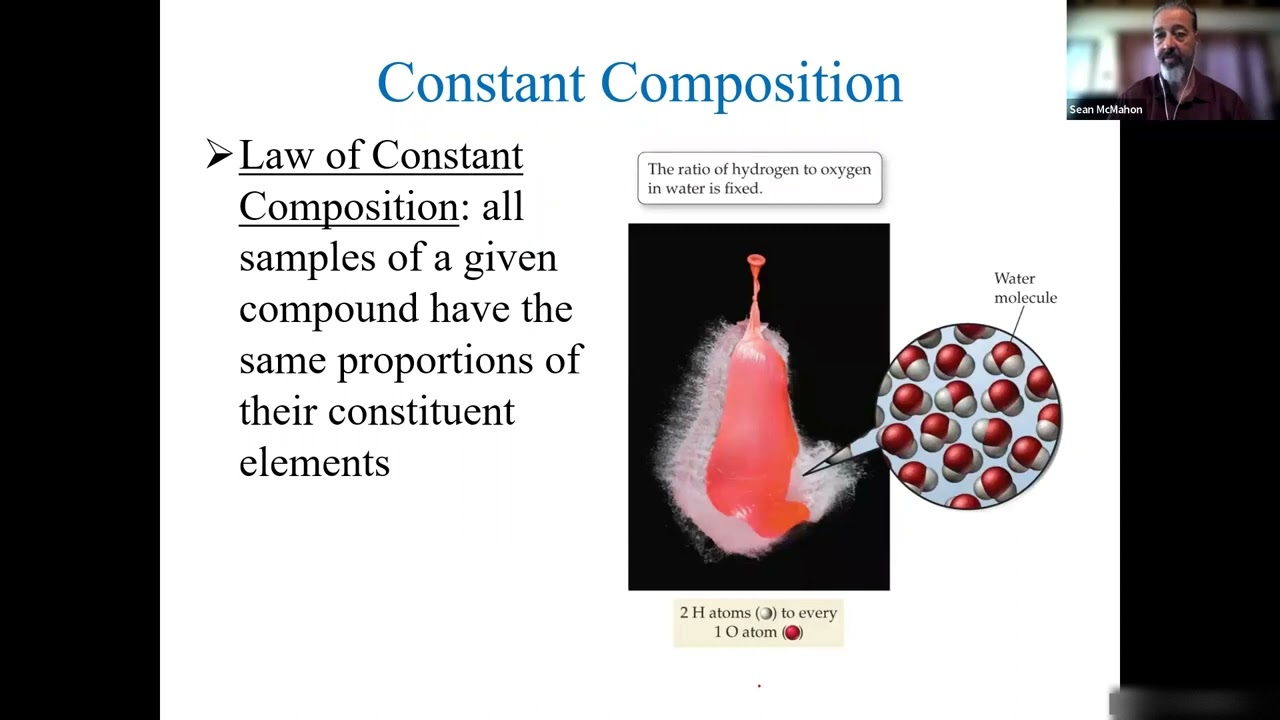
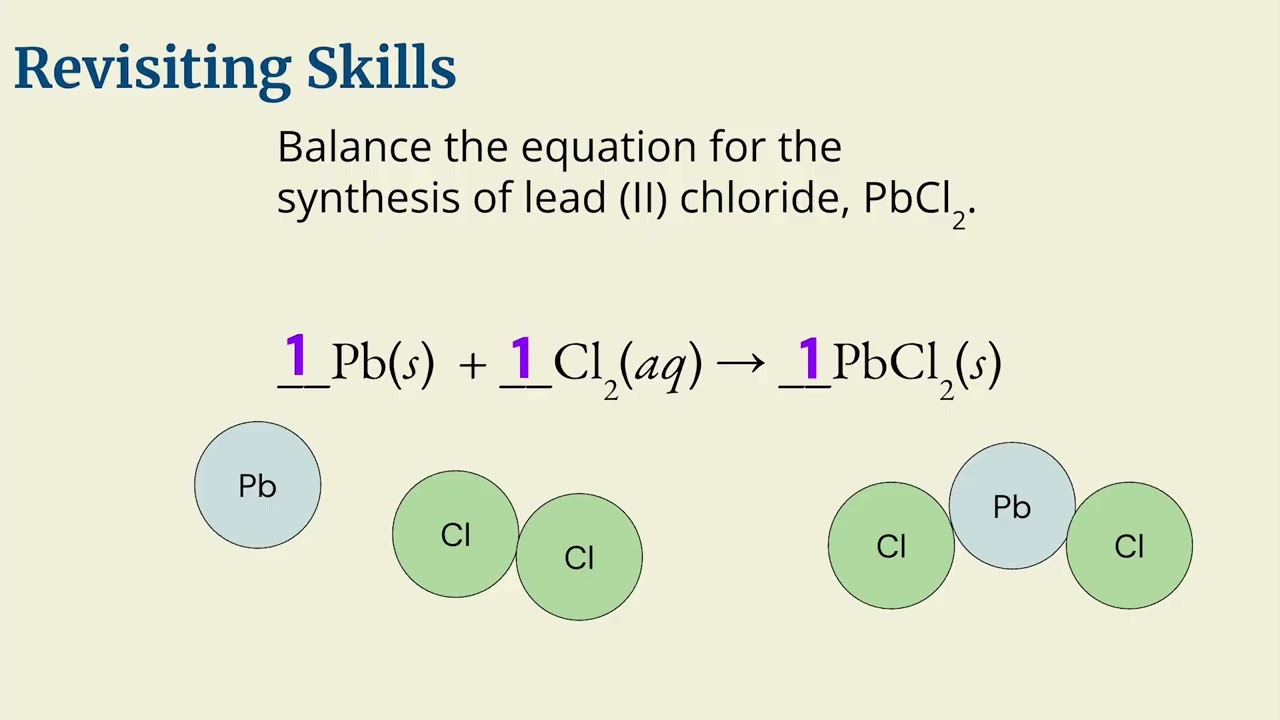
Chapter 8: Lecture 1 - Moles and Basic Stoichiometry
Автор: Science, Math, and Chemistry
Загружено: 2025-01-09
Просмотров: 44
Описание:
Topics Covered in this Video:
0:00:00 This video explains the concept of a mole (Avogadro's number)
0:09:15 We calculate Molar Mass (g/mol) for Molecules
0:14:23 We use Molar Mass (g/mol) to convert grams to moles and vice versa
0:21:52 We introduce Part 2 Converting grams to molecules using Molar Mass (2 step problems)
0:27:02 Calculating the # of atoms from Mass and vice versa
0:37:34 Students learn Stoichiometry Calculations following these Basic Rules
1. You need a Balanced Chemical Equation
2. Convert given units to the given moles
3. Convert givens moles to the desired compounds moles (mol-mol ratio)
4. Convert the desired compounds moles into the desired compounds units (g A--mol A--mol B--g B)
0:42:33 We solved mol to mol, mol to gram, and gram to gram Stoichiometric Problems together.
I apologize, but there is a careless typo in this lecture at the 48 minute mark. I'm attaching the correct answer for Exercise 13 b) here. The correct answer = 0.625 moles of octane. Thanks for your patience!
Повторяем попытку...

Доступные форматы для скачивания:
Скачать видео
-
Информация по загрузке: