AS 9701 Organic Chemistry Part 12 | Alkanes & Alkenes | Past Paper Questions
Автор: MEGA Lecture
Загружено: 2023-01-09
Просмотров: 2426
Описание:
To join our Free Trial Lesson, Join our Whatsapp Community Now! https://chat.whatsapp.com/CWSRaMi4i3w...
or msg at +92 323 509 4443
To purchases recorded courses with notes
https://megalecture.com/courses/ Book a FREE demo class for Oct Nov 2022 session through the link below: https://docs.google.com/forms/d/e/1FA...
Visit our website for video courses and other resources:
https://megalecture.com/courses/
14 Hydrocarbons
14.1 Alkanes
Learning outcomes
Candidates should be able to:
1 recall the reactions (reagents and conditions) by which alkanes can be produced:
(a) addition of hydrogen to an alkene in a hydrogenation reaction, H2(g) and Pt/Ni catalyst and heat
(b) cracking of a longer chain alkane, heat with Al 2O3
2 describe:
(a) the complete and incomplete combustion of alkanes
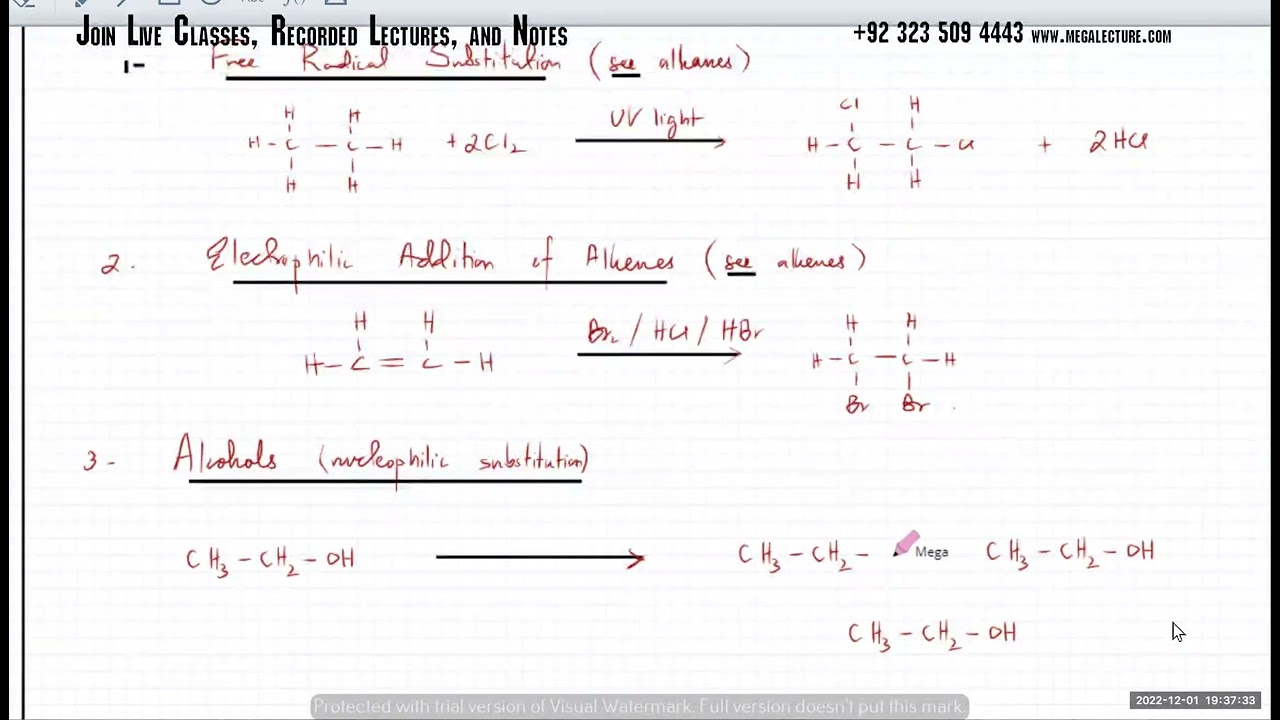
(b) the free-radical substitution of alkanes by Cl 2 or Br2 in the presence of ultraviolet light, as exemplified by
the reactions of ethane
3 describe the mechanism of free-radical substitution with reference to the initiation, propagation and
termination steps
4 suggest how cracking can be used to obtain more useful alkanes and alkenes of lower Mr
from heavier crude
oil fractions
5 understand the general unreactivity of alkanes, including towards polar reagents in terms of the strength of
the C–H bonds and their relative lack of polarity
6 recognise the environmental consequences of carbon monoxide, oxides of nitrogen and unburnt
hydrocarbons arising from the combustion of alkanes in the internal combustion engine and of their catalytic
removal
14.2 Alkenes
Learning outcomes
Candidates should be able to:
1 recall the reactions (including reagents and conditions) by which alkenes can be produced:
(a) elimination of HX from a halogenoalkane by ethanolic NaOH and heat
(b) dehydration of an alcohol, by using a heated catalyst (e.g. Al 2O3) or a concentrated acid
(c) cracking of a longer chain alkane
2 describe the following reactions of alkenes:
(a) the electrophilic addition of
(i) hydrogen in a hydrogenation reaction, H2(g) and Pt/Ni catalyst and heat
(ii) steam, H2O(g) and H3PO4 catalyst
(iii) a hydrogen halide, HX(g) at room temperature
(iv) a halogen, X2
(b) the oxidation by cold dilute acidified KMnO4 to form the diol
(c) the oxidation by hot concentrated acidified KMnO4 leading to the rupture of the carbon–carbon double
bond and the identities of the subsequent products to determine the position of alkene linkages in larger
molecules
(d) addition polymerisation exemplified by the reactions of ethene and propene
3 describe the use of aqueous bromine to show the presence of a C=C bond
4 describe the mechanism of electrophilic addition in alkenes, using bromine / ethene and hydrogen
bromide /propene as examples
5 describe and explain the inductive effects of alkyl groups on the stability of primary, secondary and tertiary
cations formed during electrophilic addition (this should be used to explain Markovnikov addition)
AS Chemistry: • AS Chemistry (New)
A2 Chemistry: • A2 Chemistry (New)
AS Physics: • AS Physics
A2 Physics: • A2 Physics
O level Physics: • O Level Physics
NET Physics: • NET Physics
AS Maths: • AS Mathematics
O level Maths: • O Level Mathematics
NET Maths: • NET Past Papers
AS Business: • Плейлист
A2 Business: • Плейлист
AS Economics: • Плейлист
A2 Economics: • Плейлист
SAT Sample Papers: • SAT Mathematics Past Papers
Повторяем попытку...

Доступные форматы для скачивания:
Скачать видео
-
Информация по загрузке: