Inert Pair Effect - class 11 - IIT JEE - NEET - IIT JAM - CSIR NET - GATE chemistry
Автор: AdiChemistry Aditya Vardhan
Загружено: 2024-09-19
Просмотров: 1207
Описание:
Today, we’ll be discussing an important topic—the Inert Pair Effect. This concept is especially useful for students preparing for exams like IIT JEE, NEET, and other competitive exams. Let’s get started!"
==============
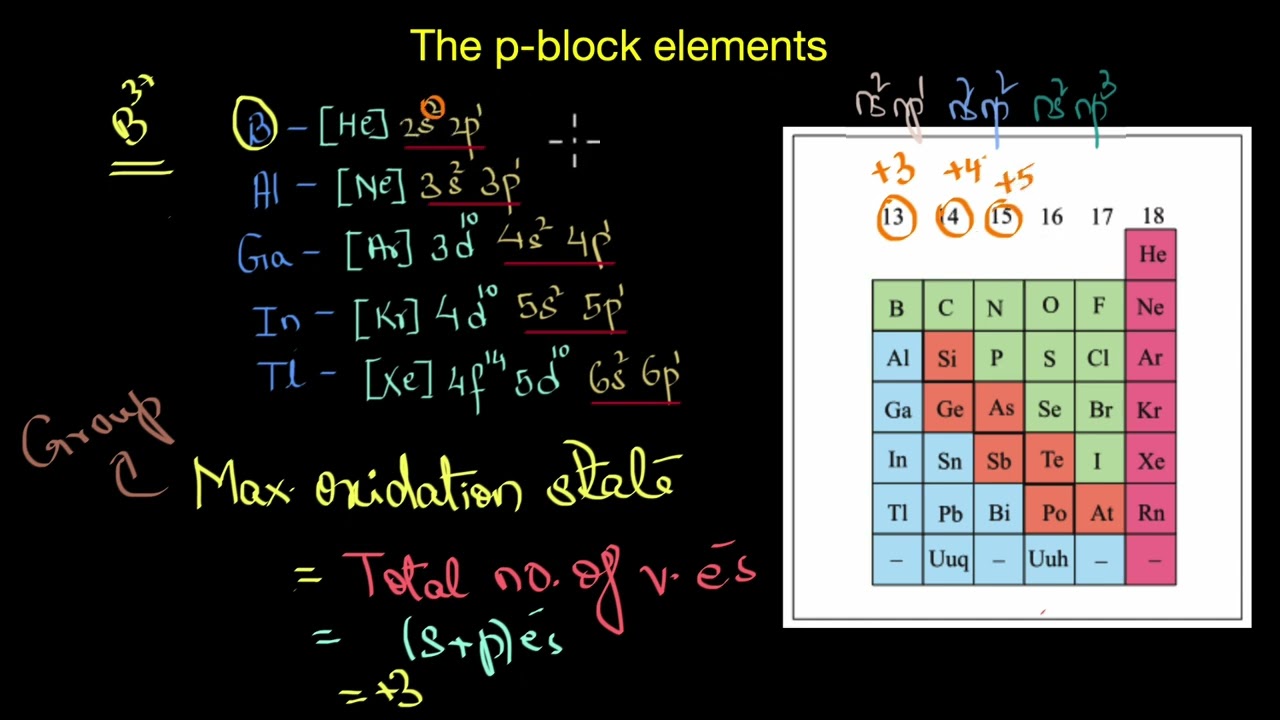
"It has been observed that some post-transition elements in the p-block, particularly those in the 4th, 5th, and 6th periods of Groups 13 to 16, tend to exhibit stable oxidation states that are two units lower than their expected group oxidation states."
"As we move down a group, the stability of the group oxidation state represented by 'n' decreases,
while the stability of the oxidation state that is two units lower than the group oxidation number , represented by (n-2) increases."
===========
"In Group 13 (Group IIIA), the typical oxidation state is +3 due to the valence configuration of ns² np¹, leading to the loss of all three electrons for stability. However, as we move down the group from aluminum to thallium, the stability of the +3 state decreases, while the +1 state becomes more stable. The heavier elements, Gallium, Indium and Thallium also show +1 oxidation state along with +3 oxidation number. Thallium is predominantly stable in the +1 oxidation state, except in compounds like its fluorides and oxides.
In thallium, the pair of electrons in the outermost 'ns' orbital tends to remain non-bonding and does not participate in bond formation, a phenomenon known as the inert pair effect. That is why, Thallium exists in +1 oxidation state rather than in +3 state.
In Group 14, the group oxidation number is +4. The valence electronic configuration is ns2 np2. However, the stability of the +4 oxidation state decreases down the group, whereas the +2 oxidation state becomes more stable. The heavier elements, Germanium, tin and lead also show +2 oxidation state. Lead, for example, is more stable in the +2 oxidation state than in the +4 state.
Once again, the two electrons in the outer ns orbital remain inert and do not participate in bond formation, illustrating the inert pair effect.
A similar trend is observed in Group 15, where the stability of the +5 oxidation state decreases as we move down the group, while the +3 state becomes more stable. Heavier elements like arsenic, antimony, and especially bismuth primarily exhibit the +3 oxidation state. Bismuth, for example, is far more stable in the +3 state, with the +5 state being relatively rare due to the increasing inert pair effect.
In Group 16, the stability of the +6 oxidation state decreases down the group, while the +4 oxidation state becomes more stable. The heavier elements like selenium, tellurium, and polonium show +4 oxidation state also. Polonium, for instance, is predominantly stable in the +4 oxidation state, with the +6 state being highly unstable due to the strong inert pair effect.
=================
Let’s define the **inert pair effect**: it is the tendency of the electrons in the outermost s-orbital to resist taking part in bond formation.
======
The question now is why the two electrons in the s orbital of the valence shell remain inert and are reluctant to take part in bond formation.
This is due to the poor shielding effect of the inner d and f orbitals, which causes the outer s-electrons to experience a stronger attraction from the nucleus. As a result, these electrons are difficult to unpair and do not engage in bond formation.
Повторяем попытку...

Доступные форматы для скачивания:
Скачать видео
-
Информация по загрузке: