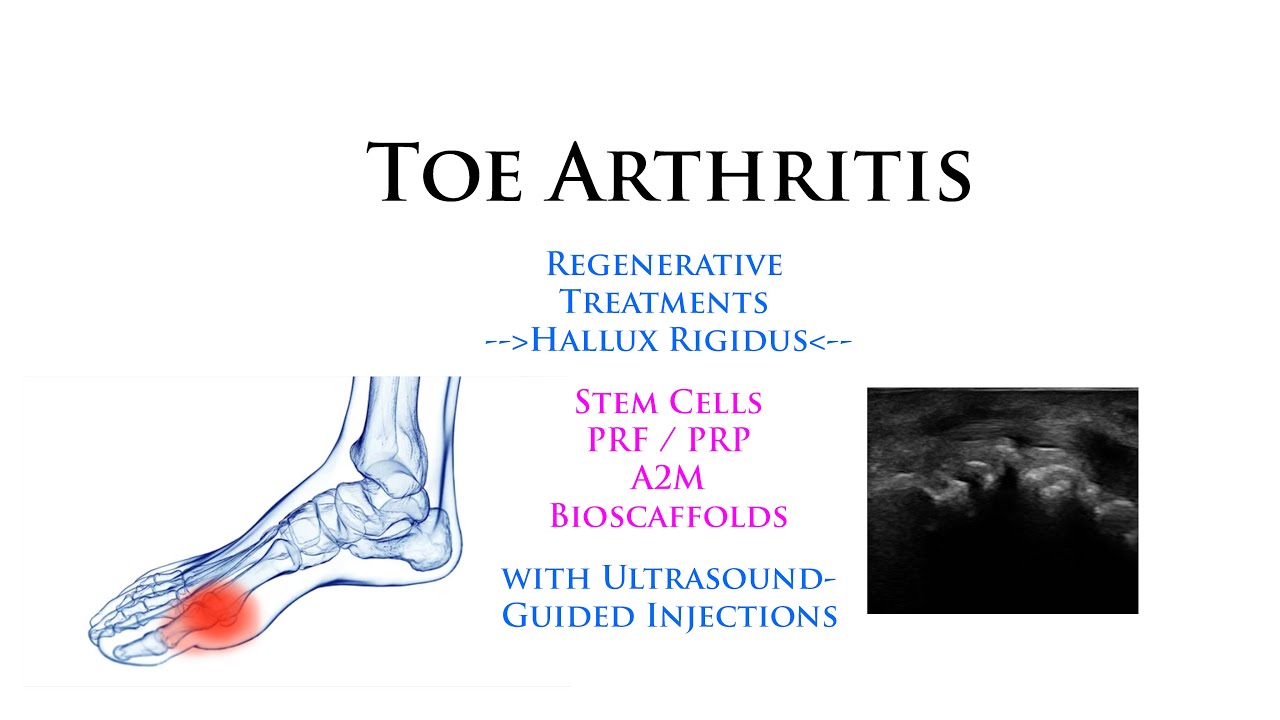
Toe Arthritis (MTP): Regenerative Stem Cells with Ultrasound-Guided Minimally-Invasive Injections
Автор: RJ McMurtrey MD MSc
Загружено: 2023-06-25
Просмотров: 268
Описание:
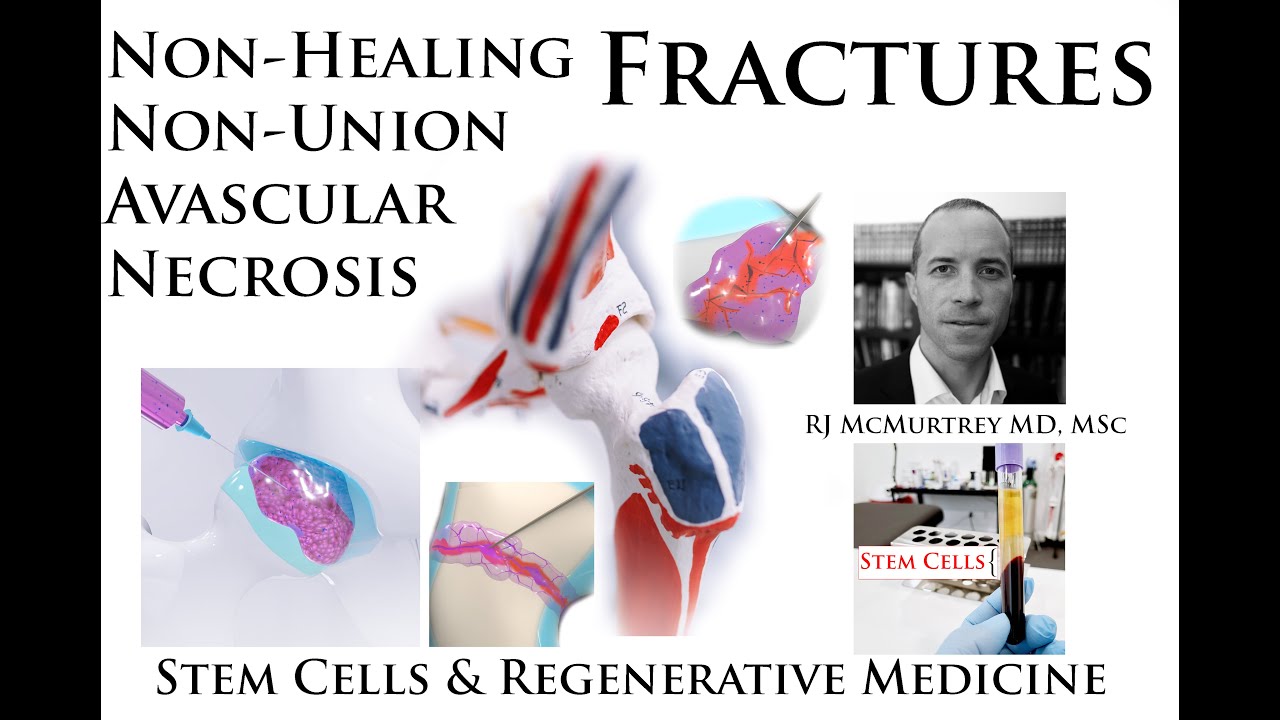
Toe Joint Arthritis - MTP Joint Injections - Regenerative Stem Cells with Ultrasound-Guided Minimally-Invasive Osteoarthritis Treatment Interventions: Many people know about arthritis in the knees and hips, but not many know about similar osteoarthritis conditions that frequently occur in the toes, fingers, spinal facet joints, and many other important joints. Fortunately I treat all of these with a variety of minimally-invasive ultrasound-guided regenerative procedures including Stem Cells, PRF, Regenerative Scaffolding Matrix, and other regenerative agents and interventions.
Copyright © 2023 Richard J. McMurtrey, MD, MSc
Alpine Spine & Orthopedics Institute
https://www.AlpineSpineOrthopedics.com/
#regenerativemedicine
#regenerativeinjections
#regenerativesportsmedicine
#regenerativeorthopedics
#regeneratecartilage
#avoidsurgery
#surgeryalternatives
#thumbarthritis
#osteoarthritis
#arthritis
#jointlubrication
#jointinjections
#stemcells
#stemcelltherapy
#stemcelltreatment
Disclaimer: We seek to always provide the highest-quality evidence-based care to our patients customized for their specific conditions, injuries, and diagnoses, which may include FDA-approved therapies as well as additional investigational, alternative, or regenerative therapies. We always discuss potential risks and benefits of all these options. The information presented here is for informational use and cites the ongoing cutting-edge research and medical advancements on these relevant topics. There are many treatments, interventions, and protocols routinely practiced in medicine and surgery which the FDA has not studied nor formally approved yet which have demonstrated overwhelming evidence of efficacy and clinical benefit, while many standard treatments and common surgeries can actually have high rates of failure and complication. The FDA does not regulate the practice of medicine but rather regulates medical marketing of devices and drugs. The FDA does not conduct clinical trials or attempt to discover new treatments, but rather requires companies or other entities to fund marketing approvals. Breakthrough technologies typically require years to decades of research work to optimize the technology and collect enough data to prove efficacy and superiority, which in some cases can optionally be submitted to the FDA if there is sufficient financial backing to market a specific product or drug. Thus the FDA has not yet studied, evaluated, or formally approved many regenerative therapies currently practiced by many of the top physicians and surgeons in the United States and around the world. Some therapies, products, or interventions may still be considered investigational or "off-label" even with substantial evidence of efficacy, and many different applications of regenerative therapies continue to be researched by our institute and other top institutions around the world. The rapid evolution and advancement of medicine demands that physicians continually update their knowledge and practice techniques to adapt to future improvements and advancing technologies. These statements have not been evaluated by the FDA, and the treatments and products presented here are for informational purposes and not guaranteed to diagnose, treat, cure, or prevent any specific disease or condition. All injuries and conditions should be formally evaluated by a knowledgeable medical professional whereby standard treatments and additional therapeutic interventions may be considered with the diagnosis and treatment plan.
Повторяем попытку...
Доступные форматы для скачивания:
Скачать видео
-
Информация по загрузке: