catalytic cycle for coupling reactions
Автор: Erland Stevens
Загружено: 2019-01-07
Просмотров: 1285
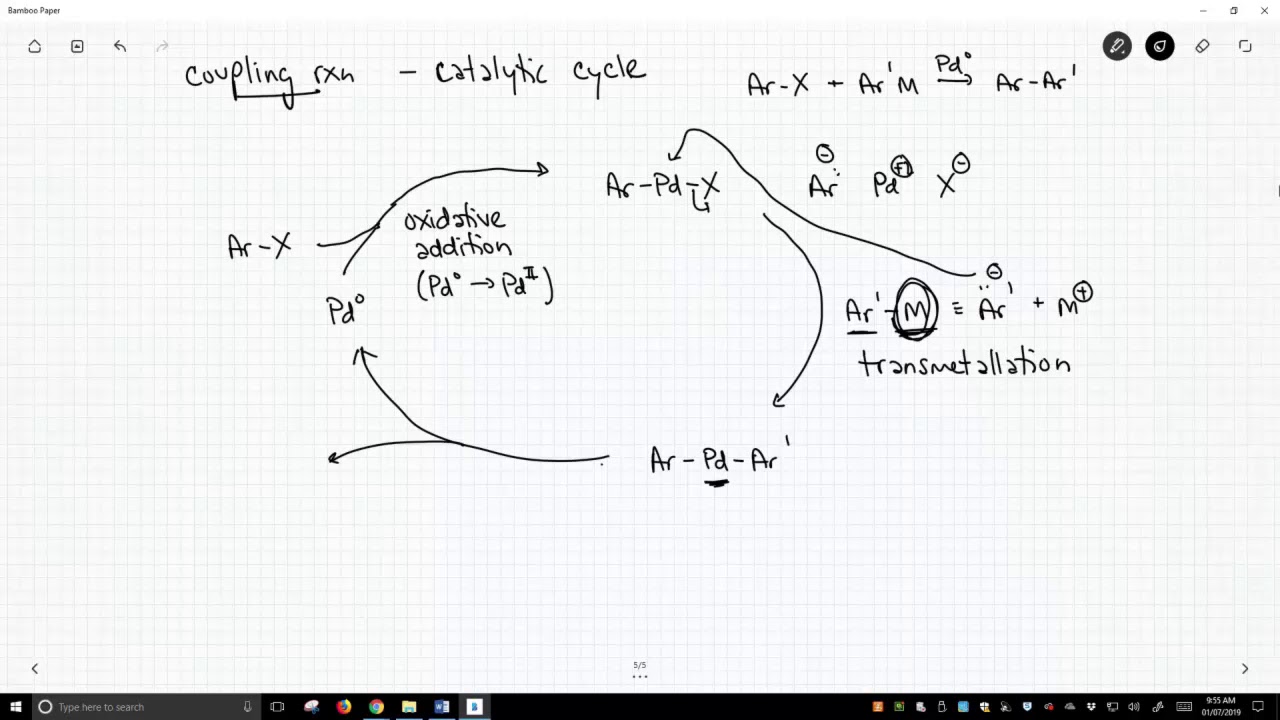
Описание: Coupling reactions that make biaryl compounds with a palladium catalyst follow a standard mechanism. The cycle begins with an aryl halide. The palladium catalyst inserts into the carbon-halogen bond to form an Ar-Pd-X species. This reaction is an example of an oxidative insertion. The Ar-Pd-X reacts with a more nucleophilic Ar'-M species, such as a Grignard or organozinc reagent, to form an Ar-Pd-Ar' species. This reaction step is called a transmetallation because the Ar' group loses its original metal (M) for a new metal (Pd). In the final step, Pd slips out of the Ar-Pd-Ar' species to form both the biaryl (Ar-Ar') and reform the palladium catalyst. This final step is called a reductive elimination because palladium is reduced from +2 to 0 oxidation state.
Повторяем попытку...
Доступные форматы для скачивания:
Скачать видео
-
Информация по загрузке: