CHEM 2325 Module 24: Amide Structure
Автор: Brian Bocknack
Загружено: 2024-06-17
Просмотров: 67
Описание:
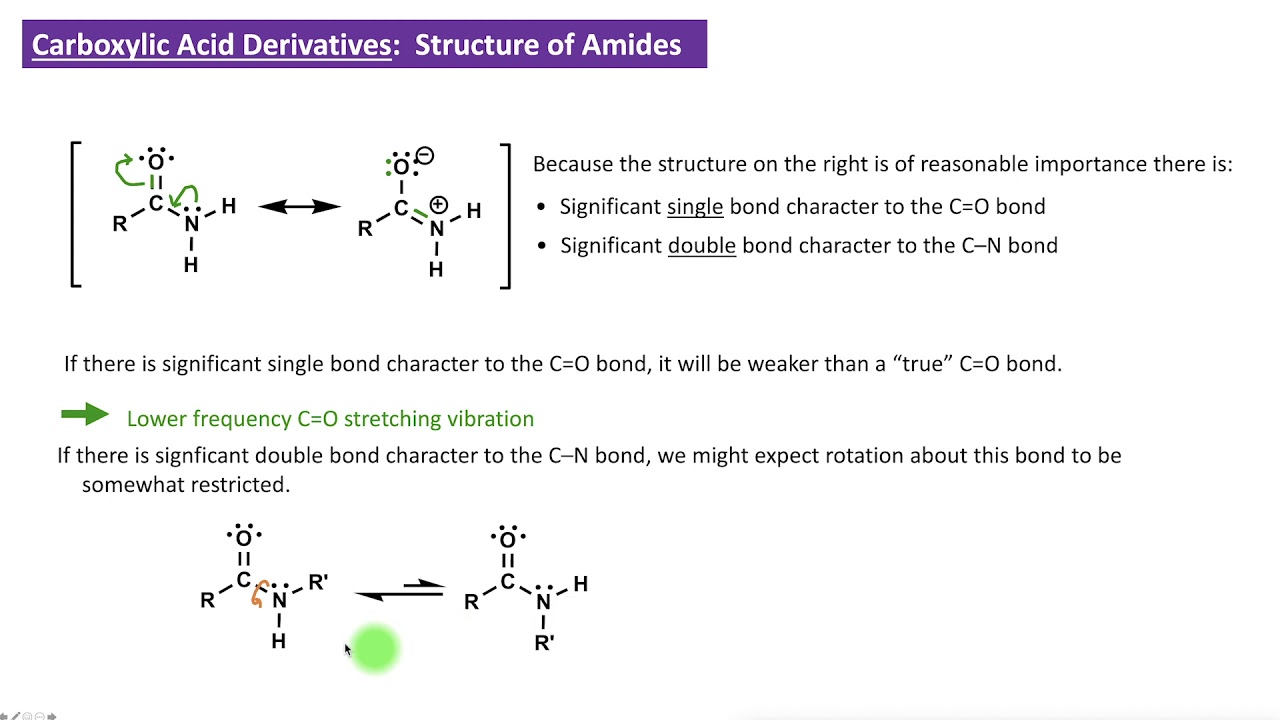
An examination of the proton NMR spectrum of the primary amide butanamide shows that the two H atoms bonded to nitrogen are not equivalent. The reason is because an amide is a resonance hybrid, and the double bond character that exists in the C-N bond restricts rotation about this bond. The fact that an amide is a resonance hybrid also explains why there is trigonal planar geometry around N, and why the N atom is not nucleophilic or basic in its reactivity.
Closed captions were generated automatically.
Повторяем попытку...
Доступные форматы для скачивания:
Скачать видео
-
Информация по загрузке:



















