🔥 Schotten Baumann Reaction Trick | Amide Ester Formation Made Easy | NEET JEE Organic Hack 😍⚡
Автор: One Chemistry
Загружено: 2026-03-01
Просмотров: 2341
Описание:
Introduction – The Reaction of Perfume Chemistry 😍
Hey toppers! 👋
Today we study the Schotten Baumann Reaction, one of the most elegant reactions in organic chemistry — widely used for preparing amides and esters.
It involves:
✔️ Acid chloride
✔️ Amine or alcohol
✔️ Base like NaOH
This reaction is used in making:
✔️ Medicines
✔️ Perfumes
✔️ Polymers like nylon
Perfect for NEET, JEE, Class 11–12, GATE, CSIR NET, UGC NET, TIFR, SET, CUTE.
Let’s make it EASY and FUN! 😍
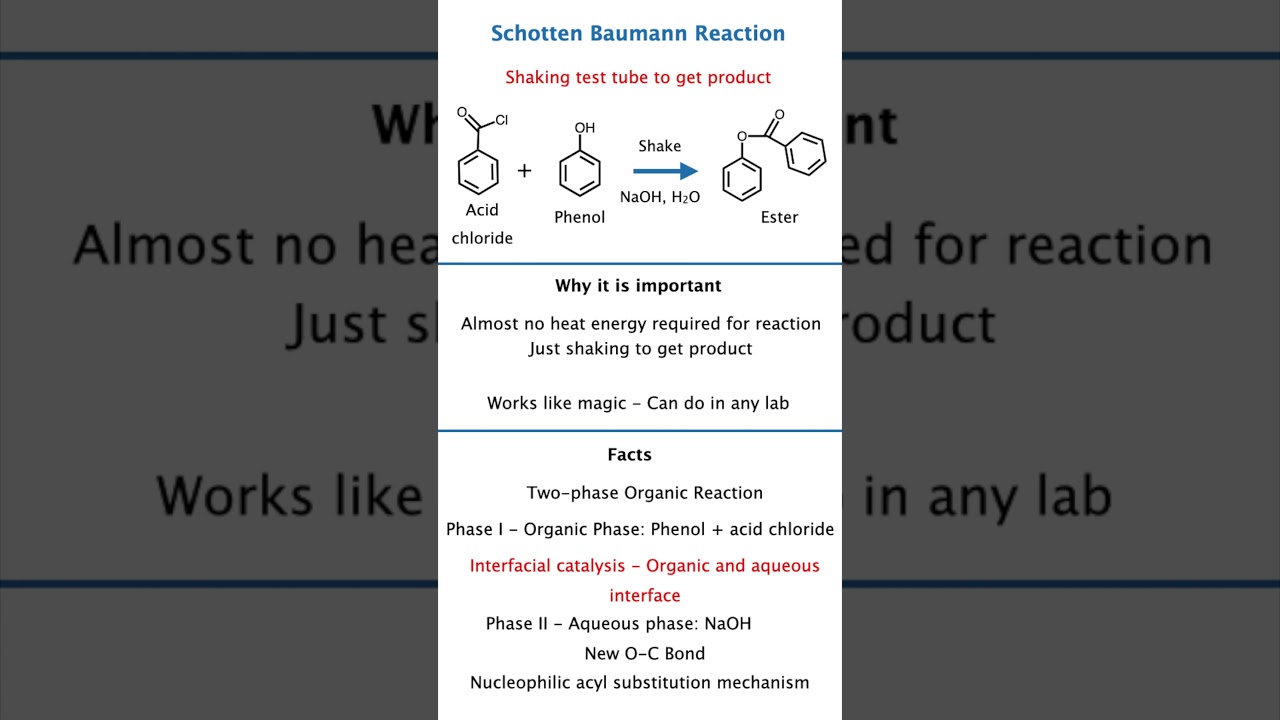
What is the Schotten Baumann Reaction? 📘
Schotten Baumann reaction is the formation of amides or esters by reacting acid chloride with amine or alcohol in the presence of a base.
Simple definition:
Acid chloride plus amine or alcohol plus base gives amide or ester.
Why Do We Need a Base? 🌟
The base (usually NaOH) neutralises HCl produced in the reaction.
Why is this important?
✔️ It prevents protonation of the amine
✔️ It speeds up the reaction
✔️ It ensures complete conversion
💬 Memory Trick:
Base removes HCl → amide forms smoothly.
Mechanism in Simple Words 🧠
1️⃣ Amine attacks acid chloride
2️⃣ Chloride leaves
3️⃣ HCl forms
4️⃣ Base neutralizes HCl
5️⃣ Final product (amide) forms
💬 Shortcut memory:
Attack → Kick out Cl → Remove HCl → Amide out!
Everyday Analogy 😄
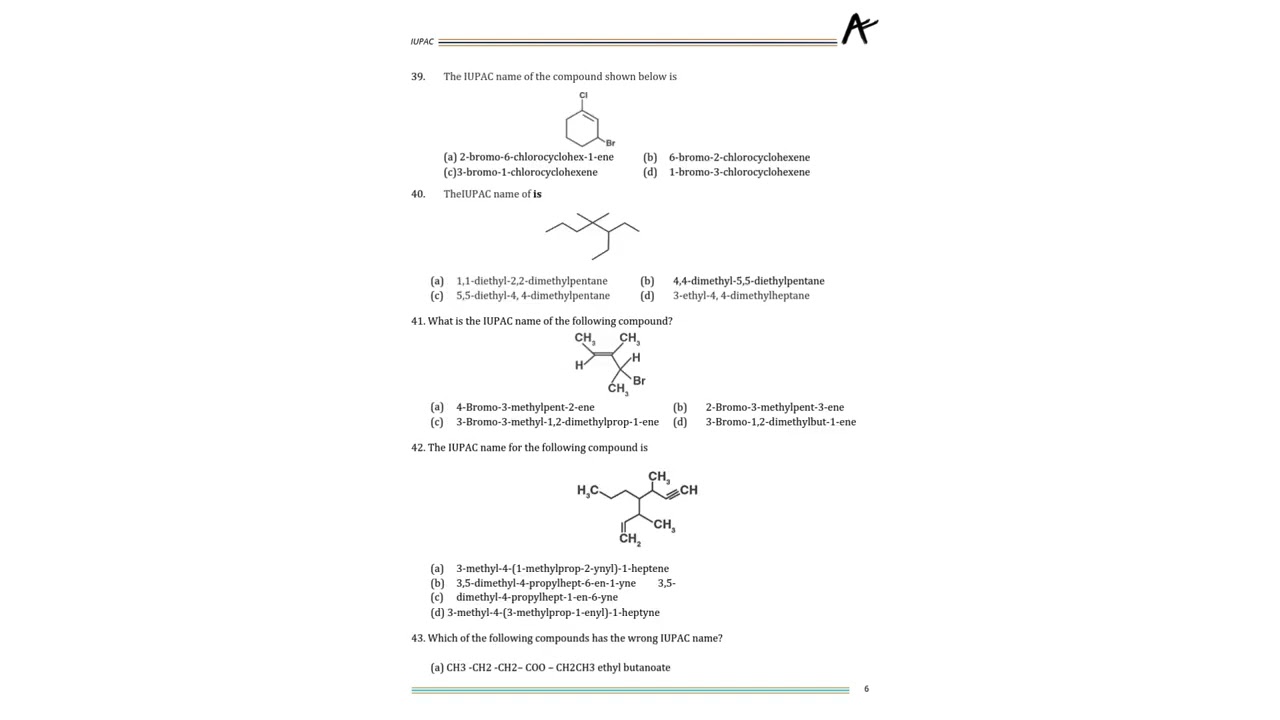
Imagine making a sandwich 🥪
The acid chloride is the bread
The amine is the filling
But to keep the sandwich neat, you wipe off extra sauce — that’s the job of NaOH 😆
That’s how Schotten Baumann works — clean and neat reaction!
Key Features YOU MUST KNOW 📝
✔️ Requires base like NaOH
✔️ Works with acid chlorides
✔️ Fast, smooth reaction
✔️ Produces amides or esters
✔️ HCl removed instantly
✔️ Very high yield
💬 One-line rule:
Acid chloride plus amine plus NaOH gives amide.
Common Examples for NEET JEE 🚀
Example 1:
Benzoyl chloride plus aniline gives acetanilide
Example 2:
Acetyl chloride plus ammonia gives acetamide
Example 3:
Benzoyl chloride plus phenol gives phenyl benzoate
💬 NEET JEE Trick:
Acid chloride always reacts faster than acid or ester.
Why is it Used in Industry? ⚡
✔️ Produces high purity amides
✔️ Works quickly
✔️ No side products
✔️ Used in perfume manufacturing
✔️ Used in polymer synthesis like nylon
💬 Master Tip:
Schotten Baumann conditions usually mean aqueous base plus acid chloride.
Exam Shortcuts, Quick Notes, High-Yield Points 🧾
✔️ Acid chloride reacts with amine
✔️ NaOH removes HCl
✔️ Amide forms
✔️ Fast reaction
✔️ Aromatic amides form easily
✔️ Used in acylation
💬 Master Code:
Schotten Baumann = amide plus base support.
Summary & Motivation 🌈
You’ve mastered the Schotten Baumann Reaction, one of the most straightforward and exam-friendly reactions in organic chemistry! 😍
Now you know:
✔️ Mechanism
✔️ Base requirement
✔️ Product formation
✔️ Industrial uses
✔️ NEET JEE shortcuts
Perfect for IIT JEE, NEET, GATE, CSIR NET, UGC NET, TIFR, CUTE, SET.
Keep learning smart — Chemistry is easy when you understand the WHY 💙
🔥 Like, Share and Subscribe for more Chemistry hacks!
🔥
#SchottenBaumannReaction #AcylationReaction #AmideFormation #NEET #JEE #IITJEE #OrganicChemistryTricks #Class12Chemistry #ChemistryMadeEasy #StudySmart #AcidChlorideReaction #GATEChemistry #CSIRNET #UGCNET #TIFRExam #SETExam #CUTEExam #AmideSynthesis #OrganicReactions #AromaticChemistry #ReactionMechanism #ChemistryShorts #NEET2026 #JEE2026 #ScienceIsFun #ChemistryHacks #ExamTricks #OrganicNameReactions #ChemistryMotivation #AcetanilidePreparation
Повторяем попытку...
Доступные форматы для скачивания:
Скачать видео
-
Информация по загрузке: