Calculate concentration of H+ from KOH solution
Автор: STEM Cat
Загружено: 2026-03-15
Просмотров: 6
Описание:
This video covers how to calculate the concentration of hydronium ions (H3O+), AKA the concentration of hydrogen ions (H+), from a solution with a known concentration of KOH.
Step 1: Calculate pOH
Step 2: Calculate pH = 14 - pOH
Step 3: Plug the pH you got into pH = -log([H3O+]) and solve for [H3O+]
Question description: Calculate [H3O+] for a 1.10 * 10^-2 M KOH solution.
Related videos:
• Acid Base Reactivity Trends | Organic Chem 1
• pH, pKa, and Predominant Protonation State...
• Equalities (Solving for x) | Middle School...
If you have any requests for other videos you want to see, please leave them down in the comments!
Thanks for watching!
Sub count: 25
Повторяем попытку...
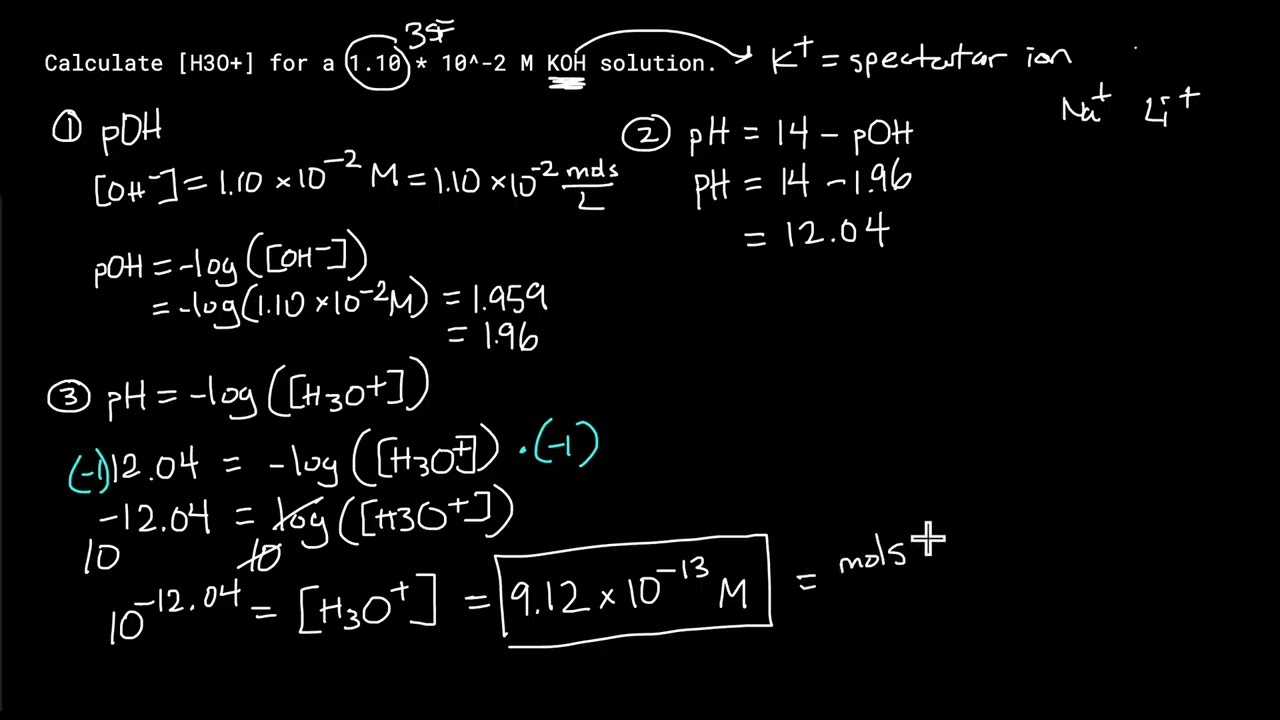
Доступные форматы для скачивания:
Скачать видео
-
Информация по загрузке:

![14.8 Lagrange multipliers to find extreme values | Multivariable Calculus [REUPLOAD]](https://imager.clipsaver.ru/ydUDVax9CtE/max.jpg)






![Эффект Джанибекова [Veritasium]](https://imager.clipsaver.ru/N9HlQ-XVnFk/max.jpg)








![16.9 Find the closed surface S using Divergence Theorem | Multivariable Calculus [REUPLOAD]](https://imager.clipsaver.ru/qAI1Njh7v3M/max.jpg)

