1 Electrochemical thermodynamics (*electrode potential, Nernst equation, etc.)
Автор: Collin Xie
Загружено: 2021-07-11
Просмотров: 4244
Описание:
Kind reminders:
(1) The lectures may best suit a student with at least a bachelor level of general physical chemistry.
(2) You may prefer a 1.25x play speed.
Major references
(which are also the strongly recommended books for learning electrochemistry):
1. R. G. Compton, C. E. Banks, Understanding Voltammetry, 3rd ed., World Scientific, New Jersey, 2018 [Currently the most recent reference book]
2. A. J. Bard, L. R. Faulkner, Electrochemical Methods: Fundamentals and Applications, 2nd ed., Wiley New York, 2001
Outline: (0:00)
Electrode potentials vs. chemical potentials: (1:05)
Origin of electrode potentials: (9:33)
Potential-determining equilibria - Nernst equation: (14:02)
Electrochemical thermodynamics based on electrode potentials (20:57)
Notes for electrochemical potentials, interfacial potential differences and electrode potentials and various kinds of 'electrode potentials': (23:08)
Повторяем попытку...

Доступные форматы для скачивания:
Скачать видео
-
Информация по загрузке:

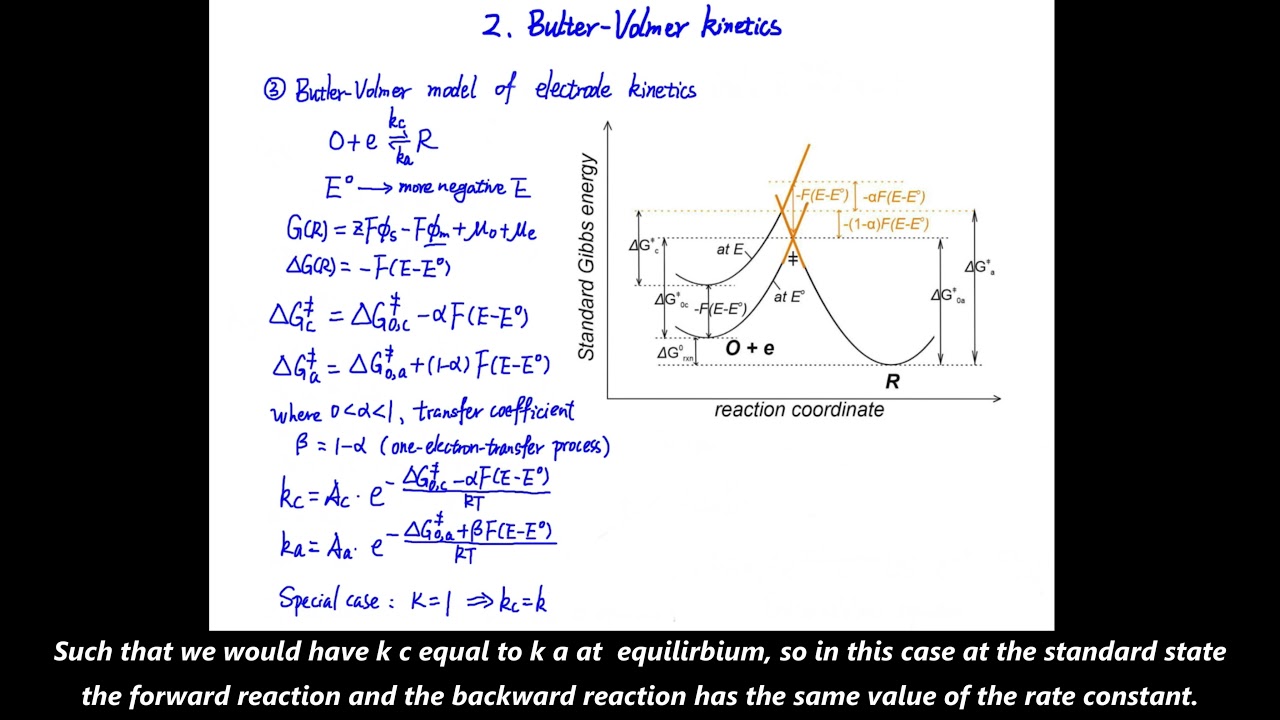
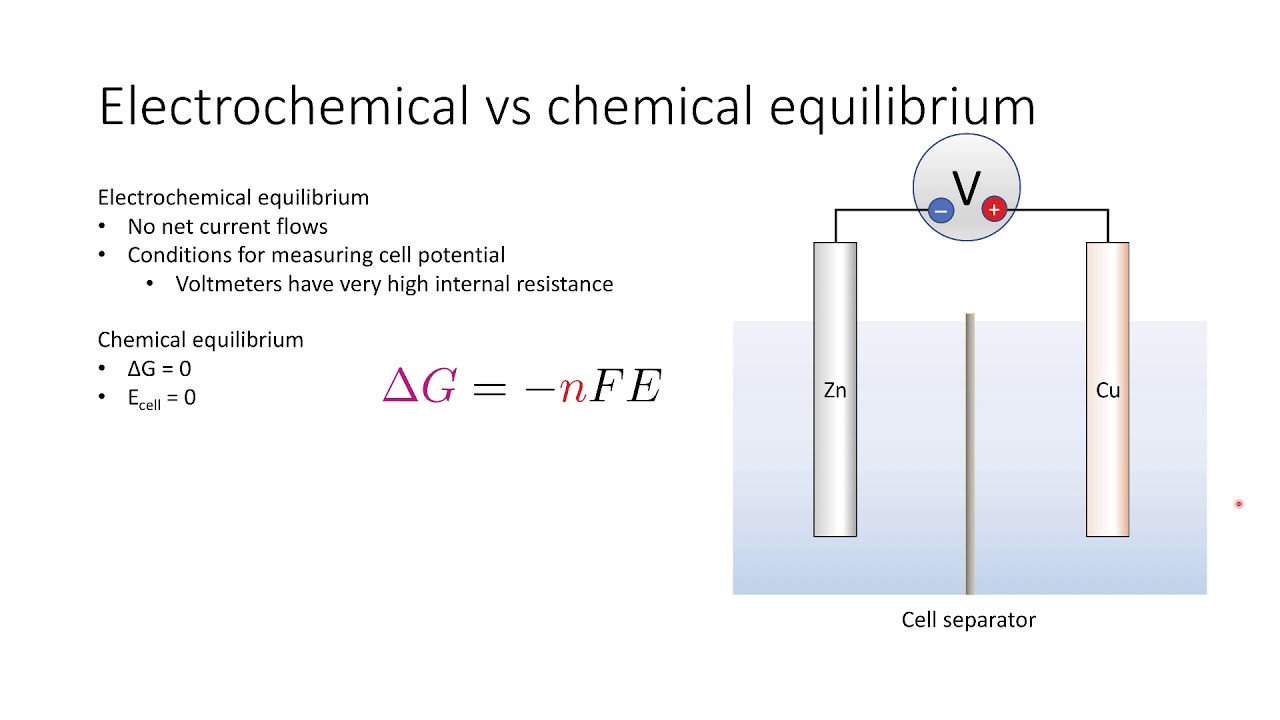
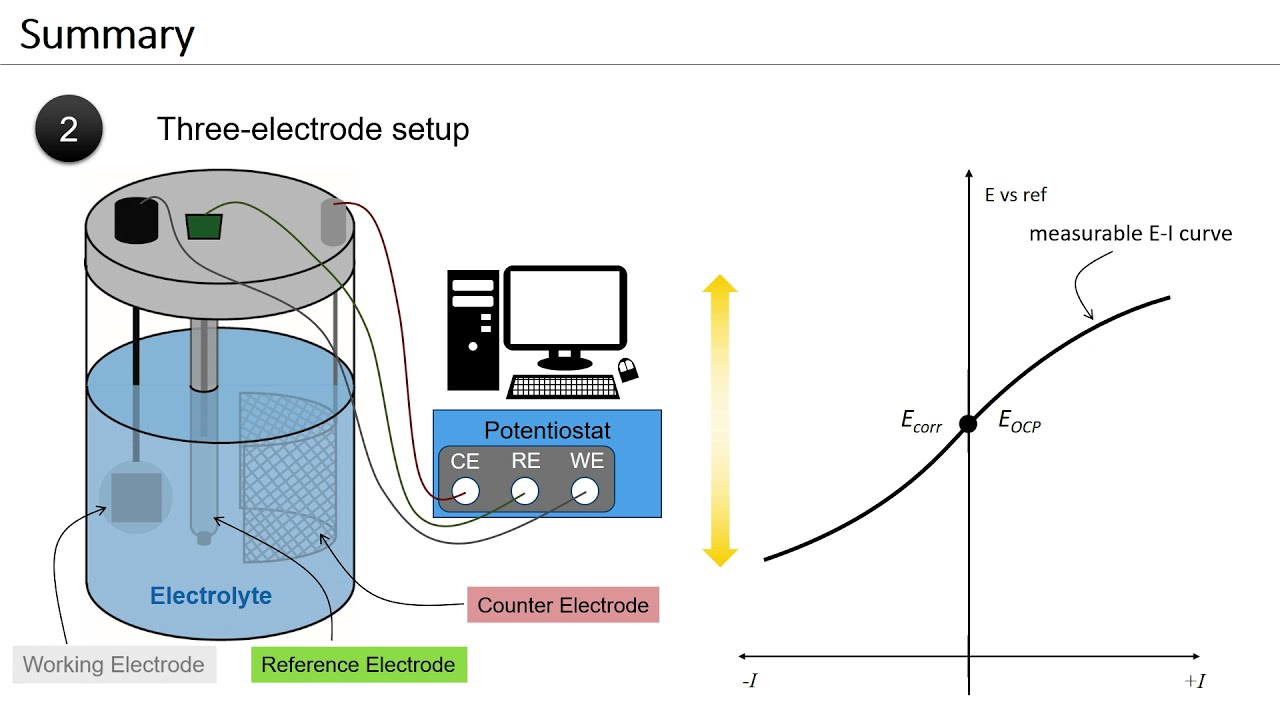












![Пожалуй, главное заблуждение об электричестве [Veritasium]](https://imager.clipsaver.ru/6Hv2GLtnf2c/max.jpg)


