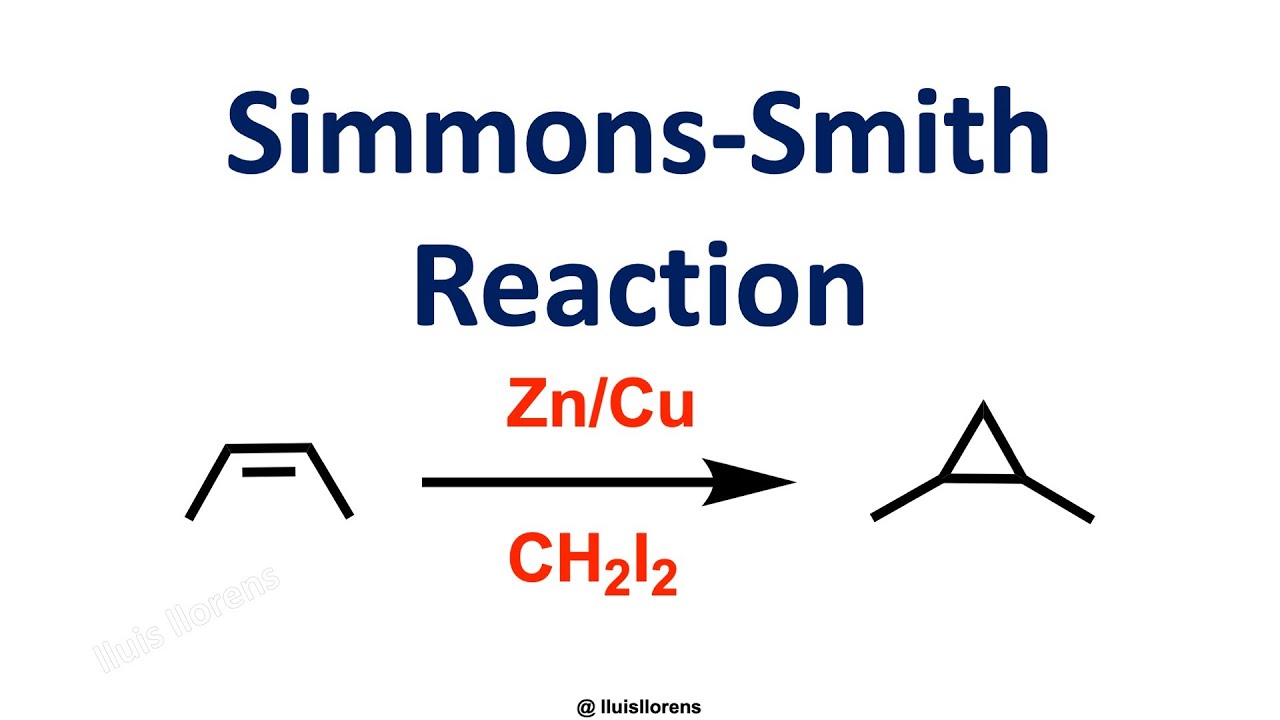
Simmons-Smith Reaction
Автор: Organic Chemistry with Lluís Llorens Palomo
Загружено: 2020-11-28
Просмотров: 18184
Описание:
The Simmons-Smith Reaction is a ring-forming reaction that allows the transformation of cyclopropanes by the reaction of alkenes with diiodomethane in the presence of zinc–copper couple.
General features:
1. A wide range of alkenes can be used: simple olefins, α,β-unsaturates ketones and aldehydes, electron-rich alkenes such as enol ethers, or enamines...
2. The cyclopropanation is stereospecific; the stereochemical information in the alkene is translated to the product.
3. Side reactions are seldom observed, and the reaction conditions are tolerant of most functional groups.
4. non-Coordinating solvents such as DCM or DCE are recommended because the use of basic solvents decreases the rate of the reaction.
Reaction mechanism:
1. Formation of the organozinc carbenoid.
2. Reaction with the alkene delivers the corresponding cyclopropane via a concerted process through a three-centered “butterfly-type” transition state.
Mechanism: https://www.pinterest.es/pin/67166973...
Pinterest:
https://www.pinterest.es/pin/67166973...
References:
https://nrochemistry.com/simmons-smit...
Seminal publications:
1. Simmons, H.E.; Smith, R.D., J. Am. Chem. Soc. 1958, 80, 19, 5323–5324. https://doi.org/10.1021/ja01552a080
2. Simmons, H.E.; Smith, R.D., J. Am. Chem. Soc. 1959, 81, 16, 4256–4264. https://doi.org/10.1021/ja01525a036
Повторяем попытку...
Доступные форматы для скачивания:
Скачать видео
-
Информация по загрузке:



















