BSc.III year QUESTION PAPER EXPLAINATION 2022 (ORGANIC CHEMISTRY) SECTION-A, PART1. @C4U CLASSES
Автор: Ashish charles
Загружено: 2022-06-24
Просмотров: 275
Описание:
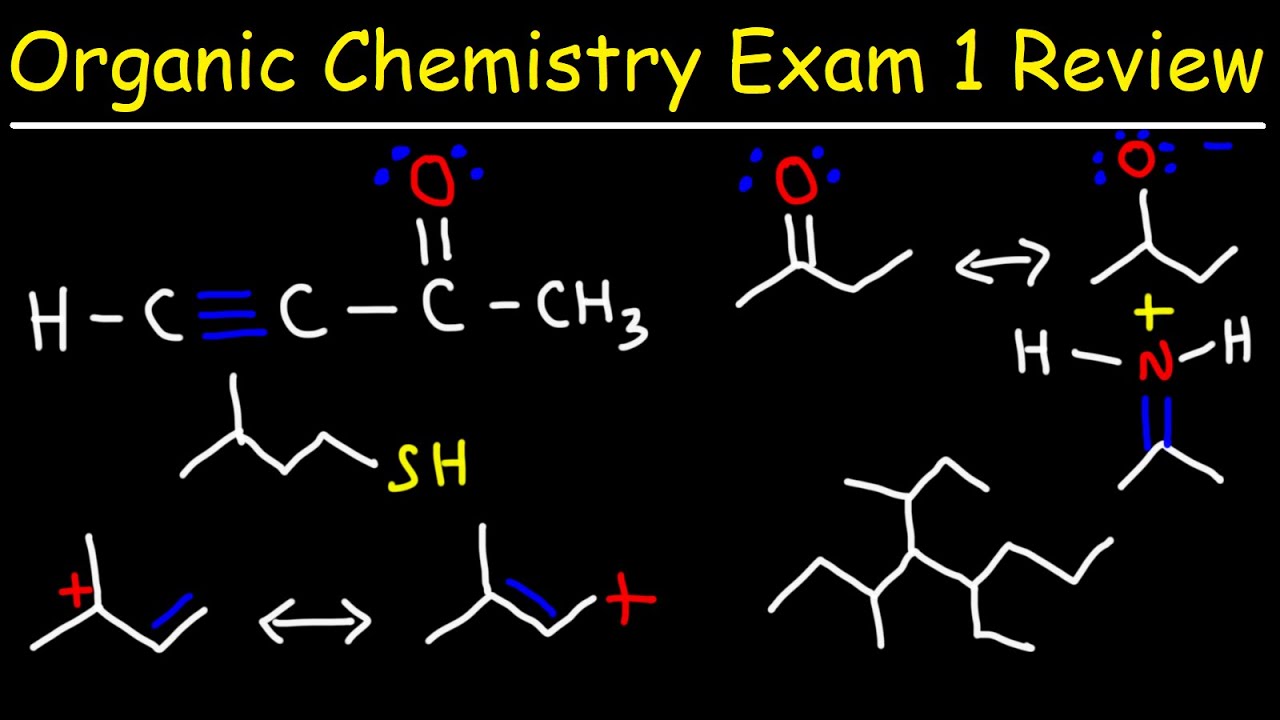
Active methylene compounds: • Active methylene compounds
Unit - III
V. Carbohydrates
Classification and nomenclature, Monosaccharides, mechanism of osazone formation, interconversion of glucose and fructose, chain lengthening and chain shortening of aldoses. Configuration of monosaccharides, Erythro and threo diastereomers, Conversion of glucose intro mannose, Formation of glycosides, ethers and esters, Determination of ring size of monosaccharides, Cyclic structure of D(+)-glucose, Mechanism of mutarotation. Structures of ribose and deoxyribose,
An introduction to disaccharides (maltose, sucrose and lactose) and polysaccharides (starch and cellulose) without involving structure determination.
VI. Amino Acids, Peptides, Proteins and Nucleic Acids:
Classification, structure and stereochemistry of amino acids, Acid-base behavior isoelectric point and electrophoresis, Preparation and reactions of O-amino acids, Structure and nomenclature of peptides and proteins, Classification of proteins, peptide structure determination, end group analysis, selective hydrolysis of peptides, classical peptide synthesis, solid-phase peptide synthesis, Structures of peptides and proteins, Levels of protein structure, Protein denaturation/ renaturation; Nucleic acids: Introduction, constituents of nucleic acids, Ribonucleosides and ribonucleotides, The double helical structure of DNA.
Unit-I
I. Spectroscopy
Nuclear magnetic resonance (NMR) spectroscopy, Proton magnetic resonance ('H NMR) spectroscopy, nuclear shielding and deshielding, chemical shift and molecular structure, spin spin splitting and coupling constants, areas of signals, interpretation of 'H NMR spectra of simple organic molecules such as ethyl bromide, ethanol, acetaldehyde, 1, 1, 2 tribromoethane, ethyl acetate, toluene and acetophenone, Problems pertaining to the structures elucidation of simple organic compounds using UV, IR and 'H NMR spectroscopic, techniques.
Unit-II
II. Organometallic Compounds
Organomagnesium compounds: the Grignard reagents, formation, structure and chemical reactions. Organozinc compounds: formation and chemical reactions.
compounds: formation and chemical reactions. Organolithium
III. Organosulphur Compounds
Nomenclature, structural formation, methods of formation and chemical reactions of thiols, thioethers, sulphonic acids, sulphonamides and Sulphaguanidine.
IV. Hetrocyclic Compounds
Introduction: Molecular orbital picture and aromatic characteristics of pyrrole, furan, thiophene and pyridine, Methods of synthesis and chemical reactions with particular emphasis on the mechanism of electrophilic substitution, Mechanism of nucleophilic substitution reaction in pyridine derivatives, Comparison of basicity of pyridine, piperidine and pyrrole.
Introduction to condensed five and six membered heterocycles, Preparation and reactions of indole, quinoline and isoquinoline with special reference to Fisher indole synthesis, Skraup synthesis and Bischler-Nepieralski synthesis, Mechanism of electrophile substitution reactions of indole, quinoline and isoquinoline.
Unit - IV
VII. Fats, Oils and Detergents
Natural fats, edible and industrial oils of vegetable origin, common fatty acids, glycerides, hydrogenation of unsaturated oils, Saponification value, iodine value, acid value, Soaps, synthetic detergents, alkyl and aryl sulphonates.
VIII. Synthetic Polymers
Addition or chain-growth polymerization, Free radical vinyl polymerization, ionic vinyl polymerization, Ziegler-Natta polymerization and vinyl polymers, Condensation or step growth-polymerization, Polyesters, plyamides, phenol formaldehyde resins, urea formaldehyde resins, epoxy resins and polyurethanes, Natural and synthetic rubbers, Elementary idea of organic conducting polymers.
IX. Synthetic Dyes
Colour and constitution (electronic Concept), Classification of dyes, Chemistry and synthesis of Methyl orange, Congo red, Malachite green, crystal violet, phenolphthalein, fluorescein, Alizarin and Indigo.
Unit - V
X. Organic Synthesis via Enolates
NMR SPECTROSCOPY PART1 • NMR(NUCLEAR MAGNETIC RESONANCE) SPECTROSCO...
• NUCLEAR MAGNETIC RESONANCE #PART2 MAGNETIC... #NMR part2
• NMR SPECTROSCOPY #PART3 MAGNETICSM & RESON... #Nmr part3
• NMR SPECTROSCOPY #PART4 #nmr part4
• NMR SPECTROSCOPY #PART5 #nmr part5
Acidity of O-hydrogens, alkylation of diethyl malonate and ethyl acetoacetate, Synthesis of ethyl acetoacetate: the condensation, Keto-enol tautomerism of ethylacetoace Alkylation of 1, 3-dithianes, Alkylation and acylation
Повторяем попытку...

Доступные форматы для скачивания:
Скачать видео
-
Информация по загрузке: