Aromatic Compounds Library 📚 | Large Rings + Shortcuts | Hückel Rule for BIG Rings 🤯 | IIT JEE NEET
Автор: One Chemistry
Загружено: 2026-02-20
Просмотров: 2628
Описание:
🌟 Why Big Aromatic Molecules Scare Students
Small rings like benzene are easy 😊
But when rings become big, students panic 😨
This video removes fear by giving you a complete aromatic compounds library, including large molecules, explained using Hückel rule (4n+2 rule) in simple English.
🔵 What Are Aromatic Compounds
An aromatic compound is a compound that is:
Cyclic (ring shaped)
Planar (flat)
Fully conjugated (continuous p orbital overlap)
Obeys Hückel rule
These compounds are extra stable (very stable).
📜 Hückel Rule Explained in Simple Words
Hückel rule says:
A compound is aromatic if it has
4n + 2 π electrons
Where n = 0, 1, 2, 3…
Possible values:
n = 0 → 2 π electrons
n = 1 → 6 π electrons
n = 2 → 10 π electrons
n = 3 → 14 π electrons
n = 4 → 18 π electrons
Think like seats 🎟️
Only special numbers allow stability.
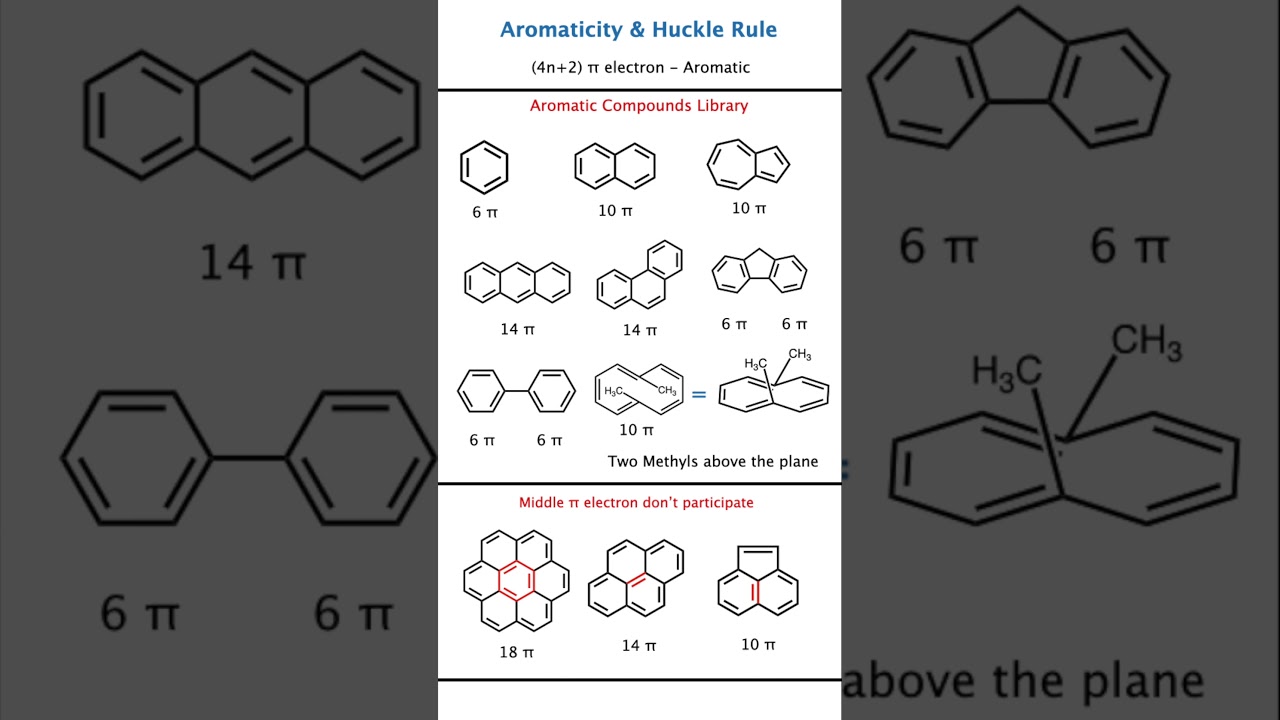
🧪 Aromatic Compounds Library – Small to BIG Molecules
1️⃣ Small Aromatic Compounds
Examples:
Benzene (6 π electrons)
Cyclopropenyl cation (2 π electrons)
Cyclopentadienyl anion (6 π electrons)
2️⃣ Medium Aromatic Compounds
Examples:
Naphthalene (10 π electrons)
Anthracene (14 π electrons)
Phenanthrene (14 π electrons)
More rings → more pi electrons → still aromatic if rule satisfied.
3️⃣ BIG Aromatic Molecules (Very Important)
Examples:
Annulenes with 10, 14, 18 π electrons
Large fused ring systems
Polycyclic aromatic hydrocarbons
Key idea:
Size does not matter, rule matters 💡
🧬 Aromatic Heterocyclic Big Molecules
Heterocyclic means ring contains nitrogen, oxygen, sulphur.
Examples:
Pyridine
Pyrrole
Furan
Thiophene
Lone pair participation decides aromaticity.
🌡️ Boiling Point Trends – Why Big Aromatic Compounds Boil Higher
Now connect with boiling point trends 🔥
Aromatic compounds generally have:
Strong pi electron cloud
Strong intermolecular attraction (attraction between molecules)
Big aromatic molecules have:
Higher molecular mass
Larger surface area
So boiling point increases with size.
Like:
Small magnet 🧲 vs big magnet 🧲🧲
📊 Factors Affecting Boiling Point (Quick Revision)
Molecular mass
Surface area
Planarity
Aromatic stability
Intermolecular force (attraction)
Big aromatic compounds usually have high boiling point.
🎯 Exam Relevance (Very High)
Asked in:
NEET Organic Chemistry
JEE Main and Advanced
Class 11 Hydrocarbons
Class 12 Aromatic Compounds
GATE Chemistry
CSIR NET
UGC NET
TIFR
CUTE
SET
⚡ Exam Tricks (Gold Level)
Count pi electrons only after checking planarity
Do not fear large rings
Use 4n+2 rule directly
🚀 One-Line Exam Shortcut
If ring is planar, conjugated and follows 4n+2 → aromatic
💡 Tips for Students
Draw structure clearly
Identify lone pair contribution
Memorise common aromatic libraries
📝 Quick Notes
Big molecules can be aromatic
Size is not a restriction
Stability comes from electron delocalisation (spreading of electrons
📌 Exam Short Notes
Naphthalene = aromatic
Anthracene = aromatic
Annulenes follow Hückel rule
Lone pair may or may not participate
✅ Summary
In this video you learned:
Meaning of aromatic compounds
Hückel rule application
Aromatic compounds library
Big molecule aromaticity
Boiling point trends
Exam shortcuts and tricks
This topic is a guaranteed scoring area 🎯
🎓 Video Suitable For
Chemistry for IIT JEE, NEET, GATE, CSIR NET, UGC NET, TIFR, CUTE, SET, and TIFR
🔖
#AromaticCompounds
#HuckleRule
#OrganicChemistry
#NEETChemistry
#JEEChemistry
#Class11Chemistry
#Class12Chemistry
#Aromaticity
#BigMolecules
#ChemistryTricks
#ChemistryShortcuts
#BoilingPointTrends
#OrganicChemistryBasics
#ScoringTopics
#NEET2026
#JEE2026
#IITJEE
#NEETAspirants
#GATEChemistry
#CSIRNET
#UGCNET
#TIFR
#CUTE
#SET
#ChemistryRevision
#LearnChemistry
#ChemistryExplained
#AromaticLibrary
#OrganicChemistryRevision
#ChemistryEasy
Повторяем попытку...
Доступные форматы для скачивания:
Скачать видео
-
Информация по загрузке:



















