How to Calculate the Rate Constant (k) Using the Arrhenius Equation | Chemical Kinetics
Автор: Learn CHEMISTRY w/Professor T.K the Chemist
Загружено: 2026-03-19
Просмотров: 3
Описание:
How to Calculate the Rate Constant (k) Using the Arrhenius Equation | Chemical Kinetics
Access worksheets and practice problems used in my LIVE sessions and full chemistry modules here:
https://tkthechemist.com/l/pdp/genera...
You can also book one-on-one chemistry tutoring and explore full chemistry courses designed for college and pre-nursing students. Go here:
https://mygemsforkids.com/chemistry-t...
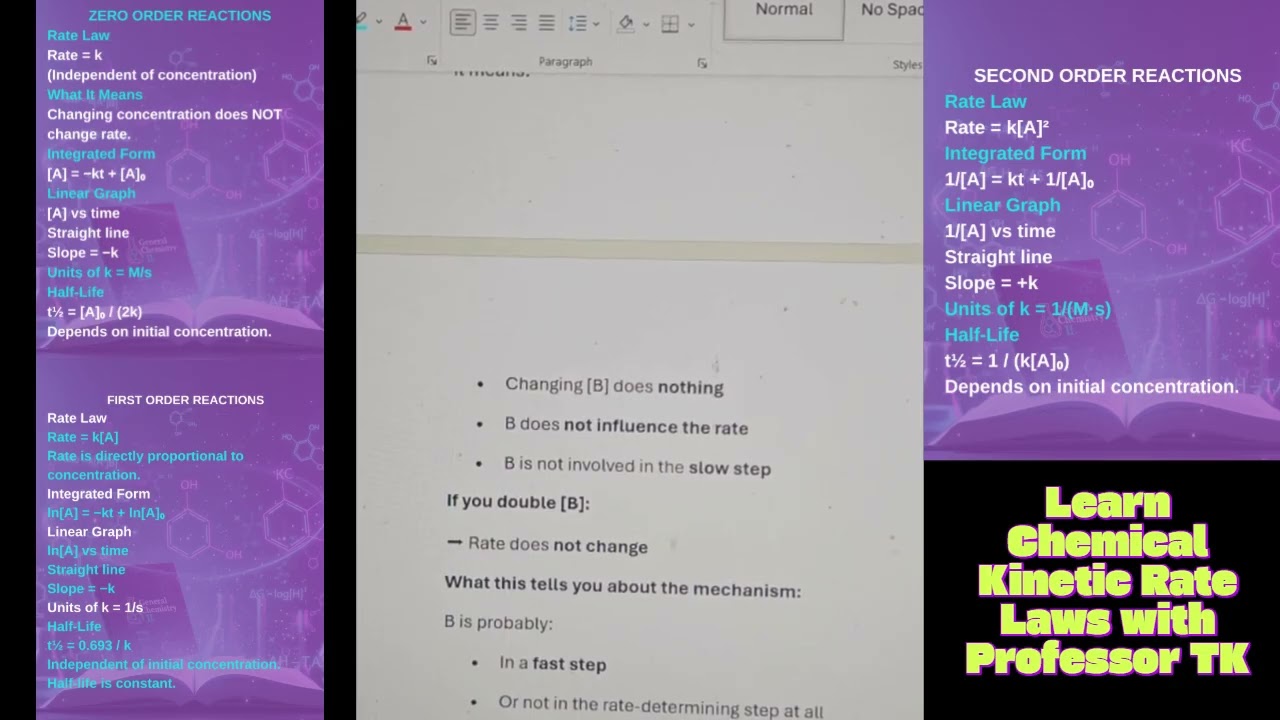
In this General Chemistry 2 lesson, Professor TK the Chemist explains how to calculate the rate constant (k) using the Arrhenius Equation. Understanding how to determine the rate constant is essential when studying chemical kinetics, reaction rates, and activation energy relationships.
The Arrhenius Equation describes how temperature and activation energy affect the speed of chemical reactions:
k = A e^(-Ea / RT)
Where:
k = rate constant
A = frequency factor
Ea = activation energy
R = gas constant
T = temperature (Kelvin)
In this video you will learn:
• What the rate constant (k) represents in chemical kinetics
• How temperature and activation energy affect reaction speed
• How to use the Arrhenius equation to solve for k step-by-step
• Strategies for solving General Chemistry II kinetics problems
This lesson is ideal for students studying:
• General Chemistry 2
• Chemical kinetics and reaction rate laws
• Activation energy and temperature relationships
• College chemistry exam preparation
If you are studying for a General Chemistry II exam or final, this tutorial will help you understand how the Arrhenius equation connects activation energy, temperature, and reaction rate constants.
#tkthechemist #chemistry #generallchemistry #chemicalkinetics #arrheniusequation #reactionrate
Повторяем попытку...

Доступные форматы для скачивания:
Скачать видео
-
Информация по загрузке: