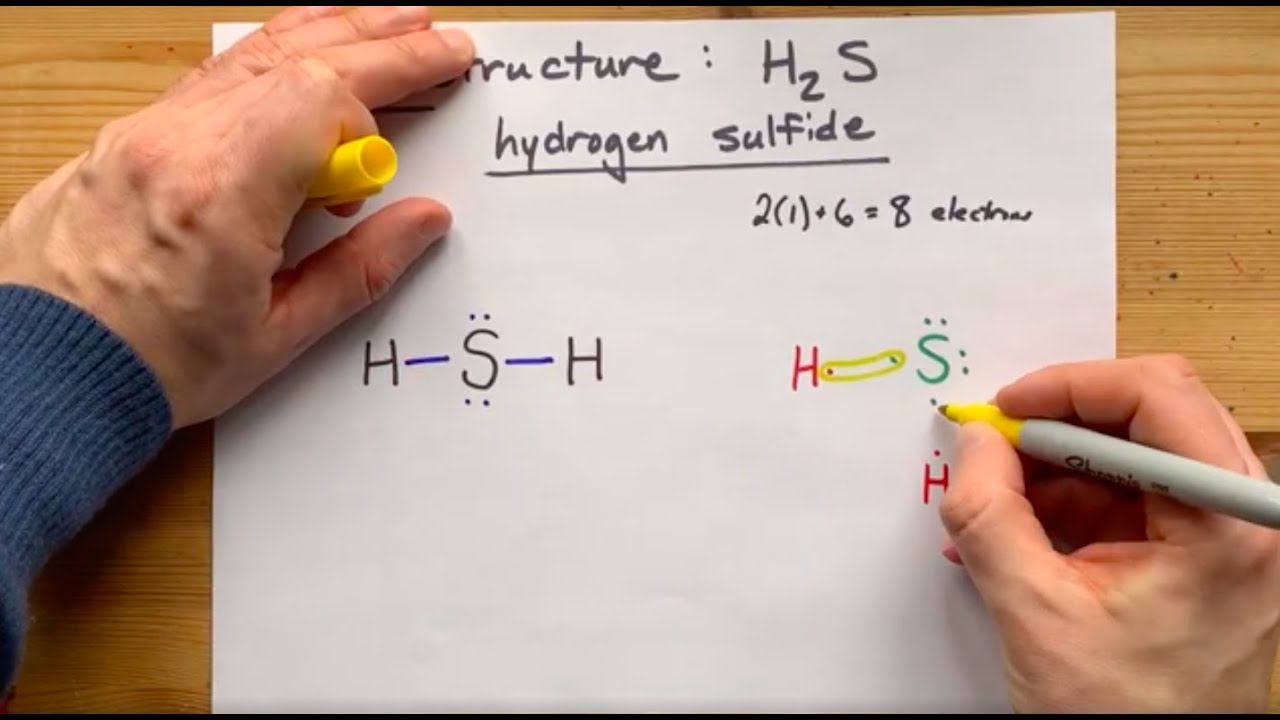
Lewis Structure of H2S, Hydrogen Sulfide
Автор: chemistNATE
Загружено: 2021-02-15
Просмотров: 60038
Описание: Hydrogen and sulfur are both non-metals, and so they SHARE electrons to form covalent bonds (this makes it a covalent aka MOLECULAR compound). Sulfur needs two extra electrons to complete its valence shell, and Hydrogen just needs one (it is an exception to the octet rule). Two single bonds are formed and the molecule is AX2E2, non-linear, bent.
Повторяем попытку...
Доступные форматы для скачивания:
Скачать видео
-
Информация по загрузке:



















