S2.2.1 Covalent bond is an electrostatic attraction [SL IB Chemistry]
Автор: Richard Thornley
Загружено: 2012-06-03
Просмотров: 55109
Описание:
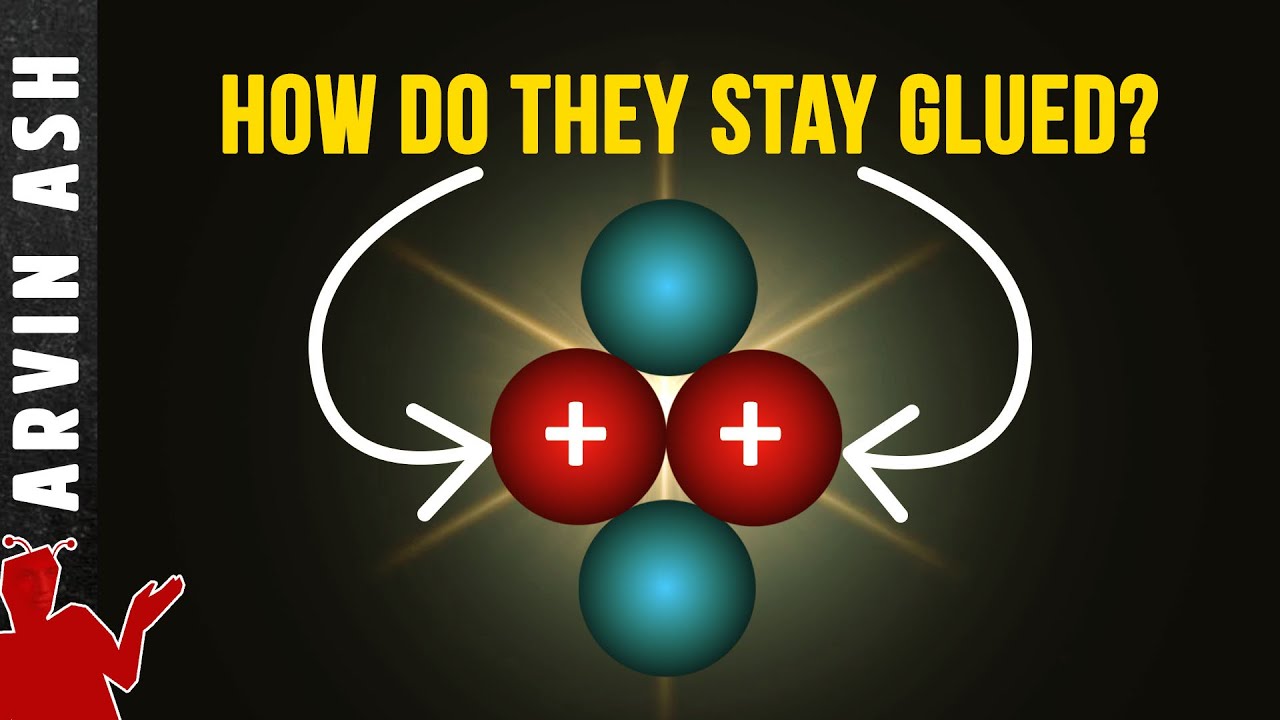
Describe the covalent bond as the electrostatic attraction between a pair of electrons and positively charged nuclei. Elements with a high electronegativity will tend to share electrons when they bond -- since they both "love" electrons.
Electrons may pair up in between the atomic nuclei to form a covalent bond. The positive nuclei have an electrostatic (opposite charges attract) attraction to the pairs. More pairs of electrons means more of an attraction and therefore stronger (multiple) bonds.
Повторяем попытку...
![S2.2.1 Covalent bond is an electrostatic attraction [SL IB Chemistry]](https://imager.clipsaver.ru/0KOQPya_WII/max.jpg)
Доступные форматы для скачивания:
Скачать видео
-
Информация по загрузке:














![Эффект Джанибекова [Veritasium]](https://imager.clipsaver.ru/N9HlQ-XVnFk/max.jpg)


![Ковалентная связь [IB Chemistry SL/HL]](https://imager.clipsaver.ru/J7hb1FdoXSs/max.jpg)
